- Review
- Open access
- Published:
The role of TDP-43 mislocalization in amyotrophic lateral sclerosis
Molecular Neurodegeneration volume 15, Article number: 45 (2020)
Abstract
Since its discovery as a primary component in cytoplasmic aggregates in post-mortem tissue of patients with Amyotrophic Lateral Sclerosis (ALS), TAR DNA Binding Protein 43 kDa (TDP-43) has remained a central focus to understand the disease. TDP-43 links both familial and sporadic forms of ALS as mutations are causative for disease and cytoplasmic aggregates are a hallmark of nearly all cases, regardless of TDP-43 mutational status. Research has focused on the formation and consequences of cytosolic protein aggregates as drivers of ALS pathology through both gain- and loss-of-function mechanisms. Not only does aggregation sequester the normal function of TDP-43, but these aggregates also actively block normal cellular processes inevitably leading to cellular demise in a short time span. Although there may be some benefit to therapeutically targeting TDP-43 aggregation, this step may be too late in disease development to have substantial therapeutic benefit. However, TDP-43 pathology appears to be tightly linked with its mislocalization from the nucleus to the cytoplasm, making it difficult to decouple the consequences of nuclear-to-cytoplasmic mislocalization from protein aggregation. Studies focusing on the effects of TDP-43 mislocalization have demonstrated both gain- and loss-of-function consequences including altered splicing regulation, over responsiveness to cellular stressors, increases in DNA damage, and transcriptome-wide changes. Additionally, mutations in TARDBP confer a baseline increase in cytoplasmic TDP-43 thus suggesting that small changes in the subcellular localization of TDP-43 could in fact drive early pathology. In this review, we bring forth the theme of protein mislocalization as a key mechanism underlying ALS, by highlighting the importance of maintaining subcellular proteostasis along with the gain- and loss-of-functional consequences when TDP-43 localization is dysregulated. Additional research, focusing on early events in TDP-43 pathogenesis (i.e. to the protein mislocalization stage) will provide insight into disease mechanisms, therapeutic targets, and novel biomarkers for ALS.
Background
TDP-43, a central player in amyotrophic lateral sclerosis
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disease characterized by the selective loss of motor neurons resulting in mortality within an average of 2-5 years [1]. Though most cases of ALS are sporadic (sALS), approximately 10% are familial (fALS) in origin. The identification of these familial cases, now spanning over 20 genes (reviewed by Nguyen et al. [2]) has highlighted the importance of various cellular processes in the pathogenesis of ALS [3]. Indeed, some rare genetic cases – such as the identification of mutations in TAR DNA Binding Protein 43 kDa (TARDBP, encoding TDP-43) have provided crucial insight into common pathogenic themes in ALS [4,5,6,7].
TDP-43 bridges the divide between sporadic and familial ALS and remains a dominant protein of interest to understand disease pathogenesis. TDP-43 was identified as a primary component of ubiquitinated and hyper-phosphorylated cytosolic aggregates observed from post-mortem tissue of patients with ALS [8, 9]. This pathological phenomena is considered a hallmark of ALS as it is observed in approximately 97% of all ALS patients regardless of the mechanisms of disease onset, with the notable exceptions of familial ALS (fALS) caused by mutations in Zn/Cu Superoxide Dismutase 1 (SOD1) and Fused in Sarcoma (FUS) [1, 10,11,12,13,14,15]. Furthermore, since the first report in 2008, over 50 mutations in TARDBP have been linked to ALS, further supporting TDP-43 dysfunction as a critical component in ALS [4,5,6, 16,17,18]. Therefore, TDP-43 dysfunction provides common ground in an otherwise convoluted disease, thus gaining notoriety and attention from researchers aiming to uncover the mechanisms causing TDP-43 aggregation. It is also important to note that mutations in TARDBP can also cause frontotemporal lobar dementia (FTLD), which itself shares some clinical parallels with ALS and displays TDP-43 pathology in ~ 45% of cases [8, 9, 19,20,21]. Here, however, we will focus on TDP-43 dysfunction as a central mechanism connecting multiple pathways in the context of ALS.
Main text
TDP-43 function, dysfunction, and aggregation
TDP-43 is a highly conserved and essential DNA/RNA binding protein belonging to the heterogenous ribonucleoprotein family that preferentially recognizes UG-rich and TG-rich motifs of RNA and DNA, respectively [22,23,24,25,26]. TDP-43 is ubiquitously expressed in all cell types and is predominantly localized to the nucleus, but is also present in the cytoplasm and mitochondria [27,28,29]. Importantly, TDP-43 is highly regulated, particularly by autoregulation through cryptic exon repression within the 3’UTR of TARDBP mRNA [30,31,32]. Deletion of TDP-43 results in embryonically lethality in mice, and its depletion or overexpression causes toxicity or cell death in cell and animal models [33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48]. Structurally, TDP-43 has a bipartite NLS sequence in the N-terminal domain upstream of the first RNA recognition motif (RRM), a nuclear export signal (NES) within the second RRM, and 5 putative mitochondria localization signals (M1-M5) of which 3 (M1, M3, and M5) are functionally characterized [14, 24, 28, 29]. The NLS and NES are important for shuttling TDP-43 between the nucleus and cytoplasm, however the involvement of the NES remains controversial as some studies suggest the NES is non-functional [27, 49,50,51]. These motifs reside within the N-terminal portion of TDP-43 forming a globular tertiary structure [22, 52, 53]. The C-terminal domain (CTD) – sometimes referred to as the low-complexity domain (LCD), glycine-rich region, intrinsically disordered region (IDR), or prion-like domain (PrLD) – remains relatively unstructured and is thought to be critically important for TDP-43 toxicity in disease [4, 53, 54]. Not only is the unstructured nature of the CTD aggregation-prone, but nearly all ALS-causing mutations on TDP-43 cluster within this domain [4, 6, 7].
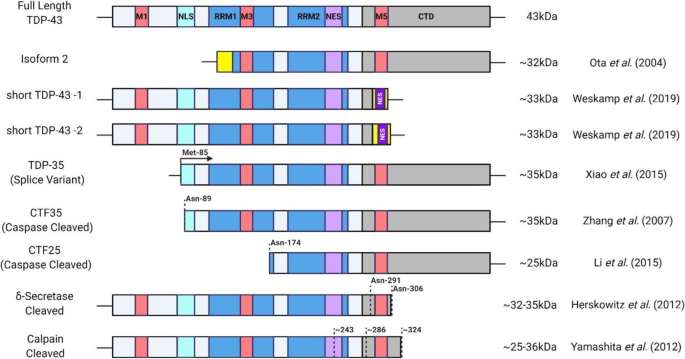
In ALS, truncated forms of TDP-43 are found in ALS aggregates, more predominantly in the cortex but also to a lesser extent in the spinal cord [55,56,57,58,59]. The N-terminally truncated, C-terminal fragments 35 kDa (CTF35) and 25 kDa (CTF25) are the most notable “species” of TDP-43 [8, 60,61,62]. Several species of TDP-43 exist and are produced through translation of alternatively spliced isoforms or through proteolytic cleavage at the post-translational level (Fig. 1). CTF35 and CTF25 can be generated through proteolytic cleavage via Caspases 3 and 7 after asparagine-89, and Caspase 4 after asparagine-174, respectively, and caspase activity is also modulated by the ALS-linked protein Progranulin (PGRN) [63,64,65,66,67,68,69]. Alternative splicing also contributes to short forms of TDP-43 where a second splice isoform was identified through cDNA sequencing encoding an N-Terminally truncated, ~ 32 kDa isoform of TDP-43 [70]. Additionally, CTF35 fragment can also be generated through non-canonical splicing in exon 2 and alternative translational initiation at methionine-85 [59]. C-terminally truncated species can also be generated through proteolytic cleavage. δ-secretase cleaves TDP-43 after asparagine-291 and -306 to generate a ~ 32 kDa and ~ 35 kDa species, respectively [71]. The calcium-dependant cysteine proteases, calpains, also play a role in TDP-43 cleavage generating ~ 35 kDa and ~ 25 kDa species associated with cell toxicity [72, 73]. As many of the truncated species of TDP-43 are of similar molecular weights many studies simply nest them as “CTF35” or “CTF25” based on molecular weight without investigation to the exact species observed which may limit the understanding of TDP-43 species contribution to ALS as different species display distinctive properties [59, 63, 74, 75]. The exact functions of these truncated species remain unclear and are generally thought to be toxic, but have also been proposed to serve a protective role in the cell to promote TDP-43 clearance [59, 63, 73, 75,76,77,78,79,80]. It is important to recognize that other species of TDP-43 CTFs have been identified at 15-16 kDa, 22-25 kDa, and 33-37 kDa in ALS/ALS-FTLD, however due to low levels of reporting their prevalence in disease remains elusive [56, 74, 75, 81,82,83,84,85].
Structure of TDP-43 including functional domains and identified short-species. M1,M3,M5 (Red): Mitochondria Localization Sequences; NLS (Turquoise): Bipartite Nuclear Localization Sequence; RRM1,RRM2 (Blue): RNA Recognition Motif; NES (Light Purple): Controversial Nuclear Export Signal; NES (Dark Purple): Nuclear Export Signal; CTD (Grey): C-Terminal Domain; Yellow Box: Alternate Amino Acid Sequence (N-Terminus of “Isoform 2” and C-Terminus of “Short TDP-43”); Dashed Lines: Cleavage Sites
There are several features that commonly define aggregates of TDP-43 in ALS. These include the accumulation of post translational modifications such as ubiquitination, poly-ubiquitination, and aberrant phosphorylation (sometimes referred to as hyperphosphorylation) of full length TDP-43; specifically phosphorylation of TDP-43 at serine 409 and 410 (S409/410) is widely used as an indicator of aggregated TDP-43 [8, 9, 56, 58, 61]. TDP-43 aggregates in ALS also accumulate full length and lower molecular weight species of TDP-43 and stain positive for the ALS-linked ubiquitin-binding autophagic adaptor Sequestosome 1 (SQSTM1, also known as p62) [8, 55,56,57,58,59,60, 82, 86,87,88,89].
The exact mechanisms mediating the formation of TDP-43 aggregates remain elusive. In ALS aggregates, TDP-43 was found to colocalize with important markers of stress granules (SGs) [90,91,92,93,94,95]. SGs are membraneless organelles that form in the cytoplasm comprised primarily of ribonuclear proteins and mRNA stalled in translation (Reviewed by Wolozin & Ivanov [96]). The formation of SGs occurs through a process called liquid-liquid phase separation (LLPS) where SG proteins and associated mRNA will de-mix into a liquid phase distinct from the cytosol [97, 98]. Two prominent proteins that are indicative of a SG are Ras GTPase-activating protein-binding protein 1 (G3BP1) and TIA1 cytotoxic granule-associated RNA binding protein (TIA1, [99,100,101,102]). Interestingly, mutations in the LCD of TIA1 – a domain that plays a key role in LLPS – cause ALS, further supporting the involvement of the cellular stress response in disease [103].
TDP-43 plays an important role in regulating the dynamics of SG formation and disassembly where loss of TDP-43 reduces SG formation [104, 105]. Treatment of cells with cell stressors used to study the formation of SGs, such as oxidative stressors (e.g. Sodium Arsenite), osmotic stressors (e.g. D-Sorbitol) or heat shock, results in the formation of phase-separated TDP-43 structures in the cytoplasm. Nevertheless, there remains a debate in the field as to whether cytoplasmic TDP-43 structures indeed colocalize as a component of SGs or are mostly distinct from these bodies [90, 91, 93, 94, 103, 104, 106,107,108,109]. Under prolonged stress conditions, phase-separated TDP-43 transitions from a liquid-like droplet to form gel-like inclusions inhibiting their ability to dissociate [110,111,112]. These gel-like inclusions eventually accumulate several hallmarks the TDP-43 inclusions seen in ALS [61, 108, 109, 111].
Clearance of TDP-43 remains an important biological process tightly coupled with cytotoxicity. The ubiquitin-proteasome system is disrupted by ALS-linked mutations in Ubiquilin-2 (UBQLN2), and is important for degrading full-length TDP-43 in addition to CTF-35 and CTF-25 [113,114,115,116,117,118,119,120]. Inhibiting this mode of clearance in primary neurons results in a greater accumulation of cytoplasmic TDP-43 aggregates compared to other cell stressors [113, 121, 122]. Recently a gain-of-function mutation in CYLD Lysine 63 Deubiquitinase (CYLD) was identified to cause ALS and FTLD [123]. The authors demonstrated in mouse primary neurons that this mutation increased deubiquitinase activity, decreased autophagy function and caused TDP-43 mislocalization, along with TDP-43 aggregation in the human brain. Autophagy also plays a role in clearing aggregated forms of TDP-43 and is linked to ALS through mutations in autophagy-related proteins SQSTM1, TANK Binding Kinase 1 (TBK1) and Optineurin (OPTN) [87, 113, 124,125,126,127,128]. Of particular importance, the sequestration of SQTSM1 into TDP-43 aggregates, one of the aforementioned hallmarks of ALS aggregates, leads to the inhibition of proteasome function in addition to autophagy, further promoting the accumulation of toxic, misfolded proteins in cells [129, 130].
The reduced clearance of aggregates can lead to another toxic gain-of-function mechanism: blocking intracellular transport. Aggregates are observed throughout the cytoplasm, often in the soma, but are also observed in the axons and dendrites [131, 132]. Inhibiting axonal transport is a common feature in ALS and particularly relevant as mutations in genes involved in cellular transport, namely KIF5A or DCTN1, cause ALS [133,134,135,136,137,138,139]. This may provide some insight into selective neuron vulnerability in ALS as motor neuron axons are particularly long and susceptible to changes in trafficking dynamics [139]. Additionally, TDP-43 plays an important role in axonal trafficking of mRNA granules, a function lost when it is mutated or aggregated [131, 132, 140,141,142].
Additional avenues of TDP-43 toxicity
Studies have suggested that not all aggregates are equal in their ability to cause toxicity. Similar to other neurodegenerative diseases, large protein aggregates such as amyloid-like structures may not be as toxic as smaller ones that preceded them such as oligomers [143,144,145,146,147]. However, describing aggregates simply as “large” or “small” is a gross oversimplification as there are thought to be multiple species of aggregates based on the properties of protein misfolding which may mediate altered toxicity at different stages [53, 147,148,149,150]. Although TDP-43 aggregation is apparent in various modes of cellular dysfunction, critics argue that TDP-43 aggregates may simply be an artifact of neuronal degeneration observed at the time of post-mortem analyses [14, 151]. In cell and animal models, TDP-43 aggregation is not necessarily essential to cause cellular toxicity [14, 152,153,154,155,156,157]. This would suggest that TDP-43 aggregates may act as a bystander alongside a cell death pathway or work in parallel with an alternate mechanism to promote toxicity. Furthermore, TDP-43 aggregates are not exclusive to motor neurons, they can also be observed in glia and muscle tissue of ALS patients and are observed to spread in a prion-like manner throughout the brain [8, 9, 15, 57, 158,159,160,161,162]. Yet in ALS, motor neurons selectively degenerate suggesting that the presence of TDP-43 aggregates may not necessarily drive cell-death. Clearly, TDP-43 aggregation is not the only feature at play.
In the presence of ALS-causing mutations, TDP-43 often demonstrates an altered nucleocytoplasmic distribution (increased cytosolic, decreased nuclear) in comparison to its wild-type counterpart [153, 163]. This may suggest that TDP-43 dysfunction can promote cytoplasmic accumulation. However, it remains difficult to differentiate whether cellular phenotypes may be caused by mislocalization or the mutation itself. Studies have exploited the NLS sequence on TDP-43 through genetic manipulation to shed light on the consequences of mislocalization independent of mutations or aggregate formation as in the cell stress models. In cellular models, expression of TDP-43ΔNLS resulted in depletion of endogenous TDP-43WT from the nucleus and promoted the formation of insoluble inclusions in the cytoplasm [29]. In a transgenic mouse model expressing human TDP-43ΔNLS under a neurofilament heavy chain promoter for brain and spinal cord expression, mice displayed a rapidly progressive motor phenotype, loss of body mass, neuromuscular denervation, and spinal motor neuron loss [155]. These mice also exhibited high levels of phosphorylated TDP-43 aggregates throughout the brain and spinal cord. Interestingly, the authors of this study describe progressive endogenous nuclear TDP-43 depletion followed by aggregate formation in the brain and to a lesser extent the spinal cord. This infers that TDP-43 mislocalization can promote nuclear depletion and is likely upstream of aggregation. Much of the toxicity, however, was attributed to the high level of transgene expression in the animal model which can function to exacerbate the effect of induced TDP-43 mislocalization by inducing cellular stress from TDP-43 overexpression.
The cellular stress model, particularly oxidative stress (e.g. via sodium arsenite treatment), to induce SGs and TDP-43 inclusions is a widely used model to study TDP-43 mislocalization and aggregation [96, 164]. Yet systemic stress makes it difficult to differentiate phenotypes associated with TDP-43 mislocalization and accumulation to general cellular stress responses. To overcome this limitation, a novel model expressed TDP-43 fused with an Arabidopsis thaliana-derived Cryptochrome 2 (CRY2) protein to allow for optogenetic instigation of LLPS [108, 109, 111, 165]. In contrast to prolonged sodium arsenite treatment, prolonged LLPS through optogenetic stimulation in wild-type conditions results in TDP-43 inclusions within the nucleus absent of ALS hallmarks including hyperphosphorylation and SQSTM1 sequestration [108]. However, prolonged induction of LLPS on TDP-43ΔNLS or mildly stressed cells inducing a mild mislocalization resulted in cytoplasmic inclusions positive for ALS-like hallmarks. Additionally this model demonstrated in vitro that there is a relatively short time course between initial induction of LLPS to aggregate formation (within hours) and inevitable cell death in less than 6 weeks [108, 111]. This suggests a slippery slope between aggregate formation and neurodegeneration, thereby inferring that therapeutically targeting the aggregation step may be too late to have substantial impact on disease progression (Fig. 2). Increasingly, the field is focusing on mechanisms outside of TDP-43 aggregation to identify early drivers of disease.
TDP-43 (Red) mislocalizes (partially or completely) from the nucleus to the cytoplasm due to genetic and/or environmental factors causing deleterious effects to the cell. Prolonged mislocalization promotes aggregation. Under physiological conditions the cell can clear small TDP-43 aggregates through proteasomal, endosomal, or autophagic degradation. Prolonged The accumulation of TDP-43 aggregates disrupts physiological functioning (e.g. sequestration of SQSTM1) thereby exacerbating pathology and promoting neuronal degeneration. Early interventions normalizing TDP-43 localization hold the potential to prevent cellular demise
The contribution of TDP-43 mislocalization to cellular toxicity in ALS
Increasing evidence suggests that nuclear-to-cytoplasmic mislocalization of TDP-43 induces toxicity through both loss- and gain-of-function mechanisms. Classic roles for TDP-43 pertain to mRNA maturation in the nucleus, specifically acting as a repressor of alternate splicing, cryptic exon splicing, and alternate polyadenylation [25, 166,167,168,169,170,171,172,173]. Loss of these functions through mislocalization or depletion have widespread deleterious effects on the cell [170, 173]. For example, recently it was discovered that loss-of-TDP-43 decreases microtubule outgrowth specifically in motor neurons through premature polyadenylation of the Stathmin2 (STMN2) transcript [167, 174]. TDP-43 is also involved in mRNA transport, a mechanism that is dysregulated within ALS, as well as local translational regulation [131, 175]. Disruption of either of these mechanisms may effectively trap TDP-43 in the cytoplasm, inhibiting its normal functions. This hypothesis is substantiated by transcriptomic evidence showing that diseased neurons and mouse models of ALS demonstrate increases in alternative splicing events, cryptic exon inclusion and alternate polyadenylated sequences [168, 176,177,178]. Recently, the CTD of TDP-43 was found to mediate its recognition of G-quadruplex structures on RNA, facilitating subcellular transport to neurites for local translation and nucleocytoplasmic trafficking [179, 180]. Interestingly, C9ORF72 hexanucleotide repeat expansion results in G-quadruplex formation, however the relationship between TDP-43 and C9ORF72 in the context of these structures has not yet been explored [181,182,183]. Additionally, RNA binding abilities of TDP-43 have been linked to TDP-43 toxicity, though some studies suggest that RNA binding is a protective mechanism [111, 184,185,186,187,188,189]. An important finding suggests that TDP-43 RNA binding regulates its solubility and lack of RNA promotes aberrant inclusions in the cytoplasm [111, 189]. Mislocalization of TDP-43 may inhibit proper RNA trafficking to the cytoplasm and subsequently promote an environment where TDP-43 is less soluble.
The nuclear functions of TDP-43 are not limited to its RNA binding functions; TDP-43 also binds DNA at TG-rich regions to regulate gene expression and exon skipping [25, 190]. For example, TDP-43 normally binds to the promoter of Vacuolar Protein Sorting 4B (VPS4B) to repress its transcription [191]. Loss of function due to mislocalization results in a loss of VPS4B repression leading to an increased interaction with the ALS-linked protein Charged Multivesicular Body Protein 2B (CHMP2B) thereby disrupting dendritic recycling-endosome trafficking and reducing ALS-linked ERB-B2 Receptor Tyrosine Kinase 4 (ERBB4) surface expression [191,192,193]. Another nuclear role for TDP-43 is in its response to genomic double stranded breaks (DSBs) which accumulate in ALS patients [163, 194,195,196,197,198,199,200]. Mislocalization of TDP-43 through an ALS-causing mutation impair the nuclear localization of DSB-repair proteins and result in the accumulation of DNA damage promoting cell death [163, 194, 201, 202]. Loss of nuclear TDP-43 can also affect chromatin accessibility leading to altered gene expression [203, 204].
Not all consequences of TDP-43 mislocalization are attributed to nuclear loss-of-function as TDP-43 has defined roles in the cytoplasm including stress granule regulation, mRNA stability, translational regulation, local synaptic RNA regulation, mRNA trafficking, microRNA regulation, and regulation of autophagy (extensively reviewed by Birsa et al. [205]). The exact consequences of increased cytoplasmic TDP-43 on these cellular functions remains largely unknown as most studies focus on protein aggregation resulting in an effective loss-of-TDP-43 function. TDP-43 is cleared through both the ubiquitin-proteasome system and lysosomal degradation pathways (highlighted above) [206,207,208]. Interestingly, TDP-43 mislocalization through overexpression or pathogenic mutations causes vacuole fragmentation, causing cellular disruption in addition to altering its own clearance [206]. Additionally, mislocalization may prime cells to respond abnormally in certain circumstances. As TDP-43 can readily undergo phase separation, the increase in cytoplasmic density biophysically promotes LLPS to occur (reviewed by Boeynaems et al. [98]). This is apparent in models of cellular stress when TDP-43 is mislocalized as there is a significant increase in the cellular stress response including rapid formation of stress granules and TDP-43 granules [109, 111, 165]. Therefore, mislocalization may sensitize the cell to respond disproportionately to a cellular stress than it may normally be less responsive to. This was recently exemplified in a study where induced pluripotent stem cell (iPSC)-derived motor neurons, but not astrocytes, with mislocalized TDP-43 showed an increase level of cell death when seeded with TDP-43 aggregates from patient tissue [209].
Although we have focused strictly on nuclear and cytoplasmic TDP-43, it is important to highlight a role for TDP-43 at mitochondria. TDP-43 misregulation through genetic manipulation of the NLS, presence of an ALS-causing mutation, or overexpression result in an increased localization to mitochondria [28]. Within the mitochondrion, mutant TDP-43 also preferentially binds mitochondria-resident mRNA, presumably causing Complex 1 disassembly through altered expression of its components [28]. Nevertheless, this finding has been debated, as studies in cell and animal models of ALS suggest that mitochondrial energetics and metabolism are unaltered [210]. The important contribution of mitochondria in ALS however remains a focus due to mutations in the mitochondrial protein SOD1 as a primary genetic cause of fALS [11]. However, it is interesting to consider that fALS caused by mutations in SOD1 rarely present with TDP-43 pathology [15].
Together, these data suggest that both loss- and gain-of-TDP-43 function mediated by nuclear-to-cytoplasmic mislocalization cause systemic cellular dysfunction in ALS. This recognition calls for a better understanding of the native subcellular functions of TDP-43 and the consequences of mislocalization independent of aggregation.
Potential mechanisms driving TDP-43 Mislocalization
It is apparent that TDP-43 mislocalization on its own is toxic and can contribute to many of the cellular characteristics observed in ALS. However, the mechanisms governing TDP-43 localization remain largely elusive. Identifying the mechanisms driving mislocalization will be crucial to identify key mechanisms that are misregulated early in disease and can be therapeutically targeted to prevent TDP-43 pathology all together (Fig. 2). These mechanisms can cover a range of biological aspects intrinsic to TDP-43 function as well as systemic cellular function [202].
As previously described, TDP-43 contains several subcellular-regulatory sequences including an NLS, the controversial NES, and mitochondrial localization sequences M1, M3, and M5. ALS-causing mutations however rarely reside within these motifs (with the exceptions of A90V in the NLS, and mutations between amino acids 294-300 in M5), and TDP-43 mislocalization exists outside of TARDBP mutations, suggesting extrinsic factors from TDP-43 govern its subcellular localization [4, 6, 28, 151, 211,212,213,214,215,216,217,218,219,220,221,222]. Understanding the contribution of these domains to TDP-43 biology remains an important step to understand disease. To this end, targeted mutagenesis of TDP-43 NLS sequence suggests that TDP-43 is actively transported into the nucleus [49]. Furthermore, knockdown of nuclear import machinery (e.g. Importin-β) impair TDP-43 nuclear localization, increasing the cytoplasmic abundance of TDP-43 [223]. Mutagenesis of the NES does not alter TDP-43 localization suggesting the NES is non-functional, however manipulation of export machinery (e.g. Exportin 1) yields conflicting results; thus there may be overlapping mechanisms of TDP-43 export [49,50,51]. Nevertheless, nuclear pore trafficking is important to some extent for normal TDP-43 localization. Perhaps unsurprisingly, in ALS, nuclear pore trafficking is disrupted, especially in cases of patients bearing mutations in TARDBP or C9ORF72 [223,224,225,226,227,228]. Aggregates of TDP-43 sequester nuclear pore proteins which would likely exacerbate TDP-43 mislocalization and accumulation into the protein aggregates [224]. In cases of C9ORF72 repeat expansion, dipeptide repeats (DPRs) generated through repeat-associated non-AUG (RAN) translation of the expanded hexanucleotide (CCCCGG) repeat blocks and disrupts the nuclear pores leading to TDP-43 pathology [229,230,231,232,233,234,235]. TDP-43 mislocalization was also shown to exacerbate RAN translation of C9ORF72 DPRs and could contribute to nuclear pore defects in conjunction or independently from DPRs [235]. This study suggests that C9ORF72 neurotoxicity may be mediated by TDP-43, and that TDP-43 mislocalization independent of C9ORF72 DPRs can disrupt nuclear pore function. Thus, nuclear pore complex disruption is an important part of TDP-43 pathogenesis, however, this mechanism may not always precede TDP-43 mislocalization. These studies warrant further investigation into mechanisms that may hinder TDP-43 translocation into the nucleus as potential aggravators of disease.
It is clear that regulation of TDP-43 is crucial for proper function, yet relatively little is known about how TDP-43 is regulated. Post translational modifications play an important role in regulating protein function (Reviewed by Buratti [236]). Along with phosphorylation at S409/410, toxic TDP-43 generally displays an overabundance of phosphate modifications leading to the general consensus that phosphorylation of TDP-43 is toxic [58, 61, 237,238,239,240,241,242,243,244,245]. However, phosphorylation may play a protective role and promote normal function within the cell [246]. For example, phosphorylation of TDP-43 at T153 and T155 by Mitogen Activated Protein Kinase Kinase (MEK) regulates TDP-43 localization to the nucleolus after heat shock, suggesting a normal maintenance role for phosphorylation in TDP-43 biology [247]. Roles for other post translational modifications such as acetylation, poly-ADP ribosylation (PARylation), oxidation, and ubiquitination have been described suggesting that post translational modifications are likely important for normal TDP-43 function and may have unappreciated roles regulating subcellular localization [112, 236, 248,249,250].
The role of TDP-43 cleavage into CTF35 and CTF25 is gaining traction as potential contributors of normal and toxic TDP-43 function. CTF35 for example assembles into SGs and plays roles in RNA processing, however, CTF25 does not localize to SGs and remains diffuse throughout the cell [251]. Cytoplasmic localization of CTF35 and CTF25 may be due to the partial and full loss of the bipartite NLS upon cleavage, respectively [59, 63, 252]. Additionally, mutations in TDP-43 and CTF35 also increase mitochondrial localization to the mitochondrial matrix and intermembrane space, respectively [252]. This study highlights that full length TDP-43, not CTF35, may cause oxidative stress, in turn increasing TDP-43 cleavage and promoting mislocalization and aggregation. As these fragments are observed in TDP-43 aggregates, increased in disease, and may induce neuronal toxicity, they remain an interesting mechanism that may provide insight into TDP-43 biology and ALS [64, 77, 251,252,253]. Several studies have identified alternative spliced isoforms of TDP-43, yet few have been functionally characterized [22, 30, 254]. Recently, a C-terminally truncated alternatively spliced isoform of TDP-43 (“short TDP-43” or sTDP-43) was characterized and found to encode a functional NES within the alternative C-terminus (Fig. 1) resulting in a more cytoplasmic localization compared to TDP-43 [255]. Interestingly, sTDP-43 was upregulated in response to increased neuronal activity, induced mislocalization of endogenous TDP-43, and caused neurotoxicity. Additionally, the sTDP-43 isoform was abundant in TDP-43 aggregates from ALS patients’ spinal cord and tissue samples. Clearly, focusing exclusively on full length TDP-43 is not encompassing to understand its contribution to ALS. Further understanding the biological roles and consequences of cleaved and alternately spliced forms of TDP-43 will provide novel insight into ALS pathogenesis and aid our interpretations of TDP-43 contributions to disease.
Approaches to study TDP-43 Mislocalization to better understand ALS
As with TDP-43 aggregates, interpreting the extent of TDP-43 mislocalization in patient tissue remains challenging as observations are made post-mortem at the late stage of disease. It may be implied that mislocalization of TDP-43 has occurred where there are cytoplasmic TDP-43 aggregates, however the mechanism(s) of biogenesis of this pathogenic hallmark remain(s) elusive. Future studies should systematically analyze the extent of TDP-43 mislocalization in addition to aggregation in human tissue to gain a better understanding of TDP-43 pathogenesis. Relying on key late-stage hallmarks such as phosphorylation of TDP-43 or SQSTM1 sequestration may limit the understanding of early pathogenesis and may lead to the dismissal of models that do not recapitulate late-stage pathology. Though studies have not systematically analyzed the extent of TDP-43 mislocalization throughout the central nervous system, data suggest that TDP-43 mislocalization correlates with aging in the vulnerable motor neurons of mouse spinal cord tissue [256]. Further understanding the basic biology and general extent of TDP-43 mislocalization throughout the central nervous system will help gain insight into the cell-type specific vulnerabilities to key stages of TDP-43 pathology.
There is a need for developing better models that recapitulate important aspects of ALS, including behavioural, pathological, and molecular phenotypes to further understanding of disease. Although most cases of ALS display TDP-43 proteinopathy, only some transgenic mouse models, and fewer endogenous mouse models, recapitulate TDP-43 pathology and ALS-like phenotypes (ALS mouse models recently reviewed by De Giorgio et al. [257]). An interesting exception is in mice bearing mutations in Senataxin (SETX), a poorly understood protein thought to act as a DNA/RNA helicase which cause a rare juvenile-onset form of fALS and sALS [258,259,260]. Both mouse models bearing transgenic and endogenous mutations in SETX recapitulate TDP-43 mislocalization, aggregation, and ALS-like phenotypes observed in patients [258]. Within TARDBP models, many models that display TDP-43 proteinopathy rely on overexpression approaches. With the advent of genome engineering (e.g. via CRISPR/Cas9) in animals and iPSCs, models have pushed towards studying endogenous TDP-43, moving away from the heavy reliance on transgenic and toxic overexpression models. Several knock-in mouse models expressing mutant TDP-43 display a range of phenotypes depending on the mutation, but most behavioural phenotypes are quite subtle and occur at a very-late stage [254, 257, 261, 262]. For example, the TDP-43Q331K knock-in mouse model does not display significant motor phenotype whereas the TDP-43Q331K transgenic mouse display some, but not robust, motor deficits [31, 263]. This difference may be due to synergistic effects of the TDP-43 mutation and overexpression in the transgenic model. There are several iPSC models for ALS (recently reviewed by Hawrot et al. [264]) however many of them do not recapitulate key hallmarks of ALS pathology. Neurons derived from TDP-43A90V patient iPSCs display TDP-43 mislocalization (likely due to disruption of the NLS), TDP-43M337V results in slight cytoplasmic granular staining of TDP-43 in iPSC-derived motor neurons, and mislocalization of TDP-43 is observed in TDP-43M337V patient iPSC-derived astrocytes [265,266,267]. The difficulty in recapitulating TDP-43 pathology may suggest that there are additional mechanisms contributing to ALS in conjunction with TDP-43 mutations, such as changes associated with human aging or chronic stress on the cell. Attempts to exacerbate ALS pathology or behaviour deficits in physiologically relevant models may help to elucidate more complex mechanisms driving disease.
Although protein aggregation has gained the most attention to understand ALS and identify novel therapeutic targets, studying the earlier components of TDP-43 mislocalization may provide an important level of insight into the disease onset and progression. However, studying subcellular localization comes with its own challenges ranging from limitations in technology to convoluted interpretations. TDP-43 mislocalization is primarily studied using cellular fractionation methods or microscopy-based methods. Developing and optimizing more reliable methods to quantitatively analyze TDP-43 subcellular localization will enhance our understanding of critical regulators of TDP-43. For example, identifying nuclear-, mitochondrial-, and cytosol-specific post translational modifications will allow for the generation of antibodies facilitating more rapid and quantifiable detection of mislocalized TDP-43. These would parallel the antibodies raised against phospho-S409/410 TDP-43 which function as a gold-standard for detection of TDP-43 aggregates through microscopy [61]. The generation of reliable tools will help to resolve potential issues with subjectivity and enhance reproducibility to understand and characterize the consequences of TDP-43 mislocalization. Although much weight is placed on DNA sequencing for modern diagnosis, this technique offers relatively little diagnostic ability in the cases of complex diseases such as ALS. Additionally, DNA sequencing is generally limited to germline mutations and does not account for potential mosaicism which may arise during an individual’s lifetime which could lead to ALS [268]. For example, an ALS patient was reported to express TDP-43 bearing the ALS-causing Q331K mutation specifically in spinal cord neurons, but not in the occipital lobe suggesting this ALS-causing mutation is somatic and thus not likely identified through germline genome sequencing [163]. Biomarkers serve as a potential diagnostic feature in addition to providing insight into disease progression. Current biomarker candidates such as Neurofilament Light Chain are increased in ALS patients and may provide insight into disease progression, but is non-specific to ALS and only provides foresight by about 12 months before symptoms occur [269,270,271,272,273]. As TDP-43 mislocalization is a central feature in ALS and biomarkers based on phenotypes associated with mislocalization may provide the specificity and foreshadowing required for early diagnosis.
Integrative “omic” approaches will be important to identify robust biomarkers capable of diagnosis and providing early insight into disease progression. Determining the direct consequences of TDP-43 mislocalization as they pertain to ALS remains a challenge due to the incomplete understanding of TDP-43 function and general dysfunction associated with disease. Therefore unbiased, systems-based approaches will be important to understand TDP-43 biology surrounding mislocalization. Bulk RNA sequencing has provided great insight into transcriptomic changes in ALS mediated by TDP-43. This technology, however, has limited abilities to detect subtle biologically significant changes that may be cell-type specific. Incorporating single cell RNA sequencing or methods of enriching populations of interest (i.e. flow cytometry, spatial transcriptomics) will help target cell-type specific changes affected by TDP-43 mislocalization in animal and cell models that can translate to human disease [274, 275]. As TDP-43 plays important roles in regulating alternative splicing, alternate polyadenylation, and cryptic exon inclusion, deep RNA sequencing will help identify rare toxic species of RNA that can give insight into ALS progression or lead to biomarker for early disease [276, 277]. Interestingly, mislocalization of FUS was recently identified in iPSCs derived from patients bearing mutations in Vasolin Containing Protein (VCP) in addition to spinal cords from sALS patients [278]. FUS mislocalization was suggested to occur due to binding of aberrantly retained introns, namely in the SFPQ gene. The authors further suggest FUS mislocalization may be a more common hallmark of ALS than previously recognized however more evidence is required. Proteomic approaches such as immunoprecipitation-mass spectrometry or proximity-labeling mass spectrometry (e.g. APEX Proteomics or BioID) comparing wild-type to mislocalized TDP-43 will provide insight into locale-specific interactors [224, 279, 280]. Additionally, cross analysis with proteomic data from ALS tissue may provide insight into potential toxic protein-protein interactions as a result of TDP-43 mislocalization serving as early therapeutic targets. New technologies are also focusing on the subcellular localization of RNA such as APEXseq [281,282,283]. Enhancing this technology with datasets that monitor Protein-RNA binding (e.g. CLIP-seq), could greatly enhance our understanding of which transcripts are bound by TDP-43 in various subcellular compartments by comparing nuclear, cytoplasmic, and mitochondria TDP-43-RNA interactions [284]. Integrating these systems-based approaches will help to uncover novel markers of TDP-43 mislocalization and elucidate pathways leading to cellular demise in ALS.
Conclusions
Increasing evidence suggests that TDP-43 aggregation is not a single driver of pathology in ALS. TDP-43 mislocalization plays significant roles in cellular dysfunction independently and in parallel to aggregation. Increasingly the field has begun to focus on understanding the regulatory mechanisms of TDP-43 mislocalization. To this end, as protein mislocalization is likely more readily reversible than protein aggregation, understanding the mechanisms regulating TDP-43 subcellular localization will be critical for therapy development. Specifically, a better understanding of TDP-43 localization regulators will surely shed light on novel therapeutics that have the potential to be more effective earlier in disease, more generalizable to most ALS cases, and more informative biomarkers for diagnosis and analysis of progression for ALS. Lastly, given that TDP-43 pathology can also coexist with other aggregate-prone proteins, such as C9ORF72 DPRs, Tau, α-Synuclein, and poly-Q expanded Huntingtin, insight into the role of TDP-43 mislocalization in its pathogenic function will serve to better understand pathology and modes of degeneration across a spectrum of neurodegenerative diseases [184, 285,286,287,288].
Availability of data and materials
Not applicable.
Abbreviations
- ALS:
-
Amyotrophic Lateral Sclerosis
- C9orf72:
-
Chromosome 9 Open Reading Frame 72
- CHMP2B:
-
Charged Multivesicular Body Protein 2B
- CNS:
-
Central Nervous System
- CRY2:
-
Cryptochrome 2
- CTD:
-
C-Terminal Domain
- CTF25:
-
[TDP-43] C-Terminal Fragment 25 kDa
- CTF35:
-
[TDP-43] C-Terminal Fragment 35 kDa
- CYLD:
-
CYLD Lysine 63 Deubiquitinase
- DCTN1:
-
Dynactin 1
- DSB:
-
Double Stranded Breaks
- ERBB4:
-
RB-B2 Receptor Tyrosine Kinase 4 (ERBB4)
- fALS:
-
Familial ALS
- FTLD:
-
Frontotemporal Lobar Dementia/Frontotemporal Dementia
- FUS:
-
Fused in Sarcoma
- G3BP1:
-
Ras-GTPase Activating Protein-Binding Protein 1
- iPSC:
-
Induced Pluripotent Stem Cell
- KIF5A:
-
Kinesin Heavy Chain Isoform A
- LCD:
-
Low-Complexity Domain
- LLPS:
-
Liquid-Liquid Phase Separation
- M1-M5:
-
[TDP-43] Mitochondrial Localization Sequence 1-5
- MEK:
-
Mitogen Activated Protein Kinase Kinase
- NES:
-
Nuclear Export Signal
- NLS:
-
Nuclear Localization Signal
- OPTN:
-
Optineurin
- PARylation:
-
Poly-ADP Ribosylation
- PGRN:
-
Progranulin
- PrLD:
-
Prion-Like Domain
- RAN:
-
Repeat-Associated Non-AUG (translation)
- RRM:
-
RNA Recognition Motif
- sALS:
-
Sporadic ALSSG
Stress Granule
- SOD1:
-
Zn/Cu Superoxide Dismutase 1
- SFPQ:
-
Splicing Factor Proline-, and Glutamine-Rich
- SQSTM1:
-
Sequestosome-1
- sTDP-43:
-
Short (Alternatively-Spliced) TDP-43
- STXN:
-
Senataxin
- TARDBP :
-
TAR DNA-Binding Protein 43 kDa (Gene)
- TBC1D1:
-
TBC1 Domain Family Member 1
- TBK1:
-
TANK Binding Kinase-1
- TDP-43:
-
TAR DNA-Binding Protein 43 kDa (Protein)
- TIA1:
-
TIA1 Cytotoxic Granule Associated RNA Binding Protein
- UBQLN2:
-
Ubiquilin-2
- VCP:
-
Vasolin-Containing Protein
- VPS4B:
-
Vacuolar Protein Sorting 4B
- WT:
-
Wild Type
References
Hardiman O, Al-Chalabi A, Chio A, Corr EM, Logroscino G, Robberecht W, et al. Amyotrophic lateral sclerosis. Nat Rev Dis Prim. Nature Publishing Group. 2017;3:17071.
Nguyen HP, Van Broeckhoven C, van der Zee J. ALS genes in the genomic era and their implications for FTD. Trends Genet. Elsevier. 2018;34:404–23.
Chia R, Chiò A, Traynor BJ. Novel genes associated with amyotrophic lateral sclerosis: diagnostic and clinical implications. Lancet Neurol. Lancet Publishing Group. 2018;17:94–102.
Van Deerlin VM, Leverenz JB, Bekris LM, Bird TD, Yuan W, Elman LB, et al. TARDBP mutations in amyotrophic lateral sclerosis with TDP-43 neuropathology: a genetic and histopathological analysis. Lancet Neurol. NIH Public Access. 2008;7:409–16.
Pesiridis GS, Lee VMY, Trojanowski JQ. Mutations in TDP-43 link glycine-rich domain functions to amyotrophic lateral sclerosis. Hum Mol Genet. 2009;18:R156–62.
Sreedharan J, Blair IP, Tripathi VB, Hu X, Vance C, Rogelj B, et al. TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science. American Association for the Advancement of Science. 2008;319:1668–72.
Kabashi E, Valdmanis PN, Dion P, Spiegelman D, McConkey BJ, Vande VC, et al. TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis. Nat Genet. Nature Publishing Group. 2008;40:572–4.
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science. American Association for the Advancement of Science. 2006;314:130–3.
Arai T, Hasegawa M, Akiyama H, Ikeda K, Nonaka T, Mori H, et al. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun. 2006;351:602–11.
Kiernan MC, Vucic S, Cheah BC, Turner MR, Eisen A, Hardiman O, et al. Amyotrophic lateral sclerosis. Lancet (London, England). Elsevier. 2011;377:942–55.
Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, et al. Mutations in cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature. Nat Publ Group. 1993;362:59–62.
Kwiatkowski TJ, Bosco DA, LeClerc AL, Tamrazian E, Vanderburg CR, Russ C, et al. Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science. American Association for the Advancement of Science. 2009;323:1205–8.
Vance C, Rogelj B, Hortobágyi T, De Vos KJ, Nishimura AL, Sreedharan J, et al. Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6. Science. 2009;323:1208–11.
Hergesheimer RC, Chami AA, De Assis DR, Vourc’h P, Andres CR, Corcia P, et al. The debated toxic role of aggregated TDP-43 in amyotrophic lateral sclerosis: A resolution in sight? Brain. Oxford University Press. 2019;142:1176–94.
Mackenzie IRA, Bigio EH, Ince PG, Geser F, Neumann M, Cairns NJ, et al. Pathological TDP-43 distinguishes sporadic amyotrophic lateral sclerosis from amyotrophic lateral sclerosis with SOD1 mutations. Ann Neurol. Wiley-Blackwell. 2007;61:427–34.
Gitcho MA, Baloh RH, Chakraverty S, Mayo K, Norton JB, Levitch D, et al. TDP-43 A315T mutation in familial motor neuron disease. Ann Neurol. John Wiley & Sons, Ltd. 2008;63:535–8.
Buratti E. Functional significance of TDP-43 mutations in disease. Adv Genet. Academic Press. 2015;91:1–53.
Xu ZS. Does a loss of TDP-43 function cause neurodegeneration? Mol Neurodegener. BioMed Central. 2012;7:27.
Karch CM, Wen N, Fan CC, Yokoyama JS, Kouri N, Ross OA, et al. Selective genetic overlap between amyotrophic lateral sclerosis and diseases of the frontotemporal dementia Spectrum. JAMA Neurol. 2018;75(7):860–75.
Borroni B, Bonvicini C, Alberici A, Buratti E, Agosti C, Archetti S, et al. Mutation within TARDBP leads to frontotemporal dementia without motor neuron disease. Hum Mutat. John Wiley & Sons, Ltd. 2009;30:E974–83.
Kwong LK, Neumann M, Sampathu DM, Lee VMY, Trojanowski JQ. TDP-43 proteinopathy: The neuropathology underlying major forms of sporadic and familial frontotemporal lobar degeneration and motor neuron disease. Acta Neuropathol. Springer. 2007;114:63–70.
Wang HY, Wang IF, Bose J, Shen CKJ. Structural diversity and functional implications of the eukaryotic TDP gene family. Genomics. Academic Press Inc. 2004;83:130–9.
Dreyfuss G, Matunis MJ, Pinol-Roma S, Burd CG. hnRNP proteins and the biogenesis of mRNA. Annu Rev Biochem. Annual Reviews. 1993;62:289–321.
Buratti E, Baralle FE. Characterization and functional implications of the RNA binding properties of nuclear factor TDP-43, a novel splicing regulator of CFTR exon 9. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2001;276:36337–43.
Buratti E, Brindisi A, Pagani F, Baralle FE. Nuclear factor TDP-43 binds to the polymorphic TG repeats in CFTR intron 8 and causes skipping of exon 9: a functional link with disease penetrance [2]. Am J Hum Genet. University of Chicago Press. 2004;74:1322–5.
Kuo PH, Chiang CH, Wang YT, Doudeva LG, Yuan HS. The crystal structure of TDP-43 RRM1-DNA complex reveals the specific recognition for UG- and TG-rich nucleic acids. Nucleic Acids Res. 2014:4712–22 Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3985631/. Cited 2020 Jun 9.
Ayala YM, Zago P, D’Ambrogio A, Xu Y-FFY-F, Petrucelli L, Buratti E, et al. Structural determinants of the cellular localization and shuttling of TDP-43. J Cell Sci. The Company of Biologists Ltd. 2008;121:3778–85.
Wang W, Wang L, Lu J, Siedlak SL, Fujioka H, Liang J, et al. The inhibition of TDP-43 mitochondrial localization blocks its neuronal toxicity. Nat Med. Nature Publishing Group. 2016;22:869–78.
Winton MJ, Igaz LM, Wong MM, Kwong LK, Trojanowski JQ, Lee VMY. Disturbance of nuclear and cytoplasmic TAR DNA-binding protein (TDP-43) induces disease-like redistribution, sequestration, and aggregate formation. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2008;283:13302–9.
Avendaño-Vázquez SE, Dhir A, Bembich S, Buratti E, Proudfoot N, Baralle FE. Autoregulation of TDP-43 mRNA levels involves interplay between transcription, splicing, and alternative polyA site selection. Genes Dev. Cold Spring Harbor Laboratory Press. 2012;26:1679–84.
White MA, Kim E, Duffy A, Adalbert R, Phillips BU, Peters OM, et al. TDP-43 gains function due to perturbed autoregulation in a Tardbp knock-in mouse model of ALS-FTD. Nat Neurosci. Nature Publishing Group. 2018;21:1138.
Sugai A, Kato T, Koyama A, Koike Y, Konno T, Ishihara T, et al. Non-genetically modified models exhibit TARDBP mRNA increase due to perturbed TDP-43 autoregulation. Neurobiol Dis. Academic Press. 2019;130:104534.
Yang C, Wang H, Qiao T, Yang B, Aliaga L, Qiu L, et al. Partial loss of TDP-43 function causes phenotypes of amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A. National Academy of Sciences. 2014;111:E1121–9.
Schmid B, Hruscha A, Hogl S, Banzhaf-Strathmann J, Strecker K, Van Der Zee J, et al. Loss of ALS-associated TDP-43 in zebrafish causes muscle degeneration, vascular dysfunction, and reduced motor neuron axon outgrowth. Proc Natl Acad Sci U S A. National Academy of Sciences. 2013;110:4986–91.
Iguchi Y, Katsuno M, Niwa JI, Takagi S, Ishigaki S, Ikenaka K, et al. Loss of TDP-43 causes age-dependent progressive motor neuron degeneration. Brain. 2013;136:1371–82.
Wu LS, Cheng WC, Shen CKJ. Targeted depletion of TDP-43 expression in the spinal cord motor neurons leads to the development of amyotrophic lateral sclerosis-like phenotypes in mice. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2012;287:27335–44.
Wils H, Kleinberger G, Janssens J, Pereson S, Joris G, Cuijt I, et al. TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration. Proc Natl Acad Sci U S A. National Academy of Sciences. 2010;107:3858–63.
Herzog JJ, Deshpande M, Shapiro L, Rodal AA, Paradis S. TDP-43 misexpression causes defects in dendritic growth. Sci Rep. Nature Publishing Group. 2017;7:1–13.
Wegorzewska I, Bell S, Cairns NJ, Miller TM, Baloh RH. TDP-43 mutant transgenic mice develop features of ALS and frontotemporal lobar degeneration. Proc Natl Acad Sci U S A. National Academy of Sciences. 2009;106:18809–14.
Xu Y-FF, Gendron TF, Zhang Y-JJ, Lin W-LL, D’Alton S, Sheng H, et al. Wild-type human TDP-43 expression causes TDP-43 phosphorylation, mitochondrial aggregation, motor deficits, and early mortality in transgenic mice. J Neurosci. Society for Neuroscience. 2010;30:10851–9.
Fiesel FC, Schurr C, Weber SS, Kahle PJ. TDP-43 knockdown impairs neurite outgrowth dependent on its target histone deacetylase 6. Mol Neurodegener. BioMed Central. 2011;6:64.
Iguchi Y, Katsuno M, Niwa JI, Yamada SI, Sone J, Waza M, et al. TDP-43 depletion induces neuronal cell damage through dysregulation of rho family GTPases. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2009;284:22059–66.
Kabashi E, Lin L, Tradewell ML, Dion PA, Bercier V, Bourgouin P, et al. Gain and loss of function of ALS-related mutations of TARDBP (TDP-43) cause motor deficits in vivo. Hum Mol Genet. 2009;19:671–83.
Vanden Broeck L, Naval-Sánchez M, Adachi Y, Diaper D, Dourlen P, Chapuis J, et al. TDP-43 loss-of-function causes neuronal loss due to defective steroid receptor-mediated gene program switching in Drosophila. Cell Rep. Elsevier. 2013;3:160–72.
Diaper DC, Adachi Y, Sutcliffe B, Humphrey DM, Elliott CJH, Stepto A, et al. Loss and gain of Drosophila TDP-43 impair synaptic efficacy and motor control leading to age-related neurodegeneration by loss-of-function phenotypes. Hum Mol Genet. 2013;22:1539–57.
Kraemer BC, Schuck T, Wheeler JM, Robinson LC, Trojanowski JQ, Lee VMY, et al. Loss of murine TDP-43 disrupts motor function and plays an essential role in embryogenesis. Acta Neuropathol. Springer. 2010;119:409–19.
Sephton CF, Good SK, Atkin S, Dewey CM, Mayer P, Herz J, et al. TDP-43 is a developmentally regulated protein essential for early embryonic development. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2010;285:6826–34.
Wu LS, Cheng W, Hou SC, Yan YT, Jiang ST, Shen CKJ. TDP-43, a neuro-pathosignature factor, is essential for early mouse embryogenesis. Genesis. Genesis. 2010;48:56–62.
Pinarbasi ES, Caǧatay T, Fung HYJ, Li YC, Chook YM, Thomas PJ. Active nuclear import and passive nuclear export are the primary determinants of TDP-43 localization. Sci Rep. Nature Publishing Group. 2018;8:7083.
Archbold HC, Jackson KL, Arora A, Weskamp K, Tank EMH, Li X, et al. TDP43 nuclear export and neurodegeneration in models of amyotrophic lateral sclerosis and frontotemporal dementia. Sci Rep. Nature Publishing Group. 2018;8:1–18.
Ederle H, Funk C, Abou-Ajram C, Hutten S, Funk EBE, Kehlenbach RH, et al. Nuclear egress of TDP-43 and FUS occurs independently of Exportin-1/CRM1. Sci Rep. Nature Publishing Group. 2018;8:1–18.
Kuo PH, Doudeva LG, Wang YT, Shen CKJ, Yuan HS. Structural insights into TDP-43 in nucleic-acid binding and domain interactions. Nucleic Acids Res. Oxford University Press. 2009;37:1799–808.
Afroz T, Hock EM, Ernst P, Foglieni C, Jambeau M, Gilhespy LAB, et al. Functional and dynamic polymerization of the ALS-linked protein TDP-43 antagonizes its pathologic aggregation. Nat Commun. Nature Publishing Group. 2017;8:45.
Guo L, Kim HJ, Wang H, Monaghan J, Freyermuth F, Sung JC, et al. Nuclear-import receptors reverse aberrant phase transitions of rna-binding proteins with prion-like domains. Cell. Cell Press. 2018;173:677–692.e20.
Berning BA, Walker AK. The pathobiology of TDP-43 C-terminal fragments in ALS and FTLD. Front Neurosci. Frontiers Media S.A. 2019;13:335.
Tsuji H, Arai T, Kametani F, Nonaka T, Yamashita M, Suzukake M, et al. Molecular analysis and biochemical classification of TDP-43 proteinopathy. Brain. Narnia. 2012;135:3380–91.
Smethurst P, Sidle KCL, Hardy J. Review: prion-like mechanisms of transactive response DNA binding protein of 43 kDa (TDP-43) in amyotrophic lateral sclerosis (ALS). Neuropathol Appl Neurobiol. Blackwell Publishing Ltd. 2015;41:578–97.
Hasegawa M, Arai T, Nonaka T, Kametani F, Yoshida M, Hashizume Y, et al. Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Ann Neurol. Wiley-Blackwell. 2008;64:60–70.
Xiao S, Sanelli T, Chiang H, Sun Y, Chakrabartty A, Keith J, et al. Low molecular weight species of TDP-43 generated by abnormal splicing form inclusions in amyotrophic lateral sclerosis and result in motor neuron death. Acta Neuropathol. Springer Verlag. 2015;130:49–61.
Arai T, Hasegawa M, Nonoka T, Kametani F, Yamashita M, Hosokawa M, et al. Phosphorylated and cleaved TDP-43 in ALS, FTLD and other neurodegenerative disorders and in cellular models of TDP-43 proteinopathy. Neuropathology. 2010;30:170–81.
Neumann M, Kwong LK, Lee EB, Kremmer E, Flatley A, Xu Y, et al. Phosphorylation of S409/410 of TDP-43 is a consistent feature in all sporadic and familial forms of TDP-43 proteinopathies. Acta Neuropathol. NIH Public Access. 2009;117:137–49.
Jeon GS, Shim YM, Lee DY, Kim JS, Kang MJ, Ahn SH, et al. Pathological modification of TDP-43 in amyotrophic lateral sclerosis with SOD1 mutations. Mol Neurobiol. Springer US. 2018;56:1–15.
Li Q, Yokoshi M, Okada H, Kawahara Y. The cleavage pattern of TDP-43 determines its rate of clearance and cytotoxicity. Nat Commun. 2015;6:1–12.
Zhang YJ, Xu YF, Dickey CA, Buratti E, Baralle F, Bailey R, et al. Progranulin mediates caspase-dependent cleavage of TAR DNA binding protein-43. J Neurosci. Society for Neuroscience. 2007;27:10530–4.
Dormann D, Capell A, Carlson AM, Shankaran SS, Rodde R, Neumann M, et al. Proteolytic processing of TAR DNA binding protein-43 by caspases produces C-terminal fragments with disease defining properties independent of progranulin. J Neurochem. John Wiley & Sons, Ltd. 2009;110:1082–94.
Beel S, Herdewyn S, Fazal R, De Decker M, Moisse M, Robberecht W, et al. Progranulin reduces insoluble TDP-43 levels, slows down axonal degeneration and prolongs survival in mutant TDP-43 mice 11 medical and health sciences 1109 neurosciences. Mol Neurodegener. BioMed Central Ltd. 2018;13:55.
Sleegers K, Brouwers N, Maurer-Stroh S, Van Es MA, Van Damme P, Van Vught PWJ, et al. Progranulin genetic variability contributes to amyotrophic lateral sclerosis. Neurology. Lippincott Williams and Wilkins. 2008;71:253–9.
Schymick JC, Yang Y, Andersen PM, Vonsattel JP, Greenway M, Momeni P, et al. Progranulin mutations and amyotrophic lateral sclerosis or amyotrophic lateral sclerosis-frontotemporal dementia phenotypes. J Neurol Neurosurg Psychiatry. 2007;78:754–6.
Yin P, Guo X, Yang W, Yan S, Yang S, Zhao T, et al. Caspase-4 mediates cytoplasmic accumulation of TDP-43 in the primate brains. Acta Neuropathol. Springer Verlag. 2019;137:919–37.
Ota T, Suzuki Y, Nishikawa T, Otsuki T, Sugiyama T, Irie R, et al. Complete sequencing and characterization of 21,243 full-length human cDNAs. Nat Genet. Nature Publishing Group. 2004;36:40–5.
Herskowitz JH, Gozal YM, Duong DM, Dammer EB, Gearing M, Ye K, et al. Asparaginyl endopeptidase cleaves TDP-43 in brain. Proteomics. 2012;12:2455–63.
Yamashita T, Hideyama T, Hachiga K, Teramoto S, Takano J, Iwata N, et al. A role for calpain-dependent cleavage of TDP-43 in amyotrophic lateral sclerosis pathology. Nat Commun. Nature Publishing Group. 2012;3:1–13.
Rao MV, Campbell J, Palaniappan A, Kumar A, Nixon RA. Calpastatin inhibits motor neuron death and increases survival of hSOD1 G93A mice. J Neurochem. Blackwell Publishing Ltd. 2016;137:253–65.
Brower CS, Piatkov KI, Varshavsky A. Neurodegeneration-associated protein fragments as short-lived substrates of the N-end rule pathway. Mol Cell. Elsevier. 2013;50:161–71.
Igaz LM, Kwong LK, Chen-Plotkin A, Winton MJ, Unger TL, Xu Y, et al. Expression of TDP-43 C-terminal fragments in vitro recapitulates pathological features of TDP-43 proteinopathies. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2009;284:8516–24.
Suzuki H, Lee K, Matsuoka M. TDP-43-induced death is associated with altered regulation of BIM and Bcl-xL and attenuated by caspase-mediated TDP-43 cleavage. J Biol Chem. 2011;286:13171–83.
Zhang YJY-J, Xu YFY-F, Cook C, Gendron TF, Roettges P, Link CD, et al. Aberrant cleavage of TDP-43 enhances aggregation and cellular toxicity. Proc Natl Acad Sci. National Academy of Sciences. 2009;106:7607–12.
Wang X, Fan H, Ying Z, Li B, Wang H, Wang G. Degradation of TDP-43 and its pathogenic form by autophagy and the ubiquitin-proteasome system. Neurosci Lett. Elsevier. 2010;469:112–6.
Walker AK, Tripathy K, Restrepo CR, Ge G, Xu Y, Kwong LK, et al. An insoluble frontotemporal lobar degeneration-associated TDP-43 C-terminal fragment causes neurodegeneration and hippocampus pathology in transgenic mice. Hum Mol Genet. 2015;24:7241–54.
Huang CC, Bose JK, Majumder P, Lee KH, Huang JTJ, Huang JK, et al. Metabolism and mis-metabolism of the neuropathological signature protein TDP-43. J Cell Sci. Company of Biologists Ltd. 2014;127:3024–38.
De Marco G, Lomartire A, Mandili G, Lupino E, Buccinnà B, Ramondetti C, et al. Reduced cellular Ca2+ availability enhances TDP-43 cleavage by apoptotic caspases. Biochim Biophys Acta. Elsevier. 2014;1843:725–34.
Shenoy J, El Mammeri N, Dutour A, Berbon M, Saad A, Lends A, et al. Structural dissection of amyloid aggregates of TDP-43 and its C-terminal fragments TDP-35 and TDP-16. FEBS J. 2019;287:2449–67.
Osuru HP, Pramoonjago P, Abhyankar MM, Swanson E, Roker LTA, Cathro H, et al. Immunolocalization of TAR DNA-binding protein of 43 kDa (TDP-43) in mouse seminiferous epithelium. Mol Reprod Dev. John Wiley and Sons Inc. 2017;84:675–85.
YAT K, Alemu S, Lamari A, Loew N, Brower CS. The N termini of TAR DNA-binding protein 43 (TDP43) C-Terminal fragments influence degradation, aggregation propensity, and morphology. Mol Cell Biol. American Society for Microbiology. 2018;38:e00243–18.
Nonaka T, Kametani F, Arai T, Akiyama H, Hasegawa M. Truncation and pathogenic mutations facilitate the formation of intracellular aggregates of TDP-43. Hum Mol Genet. 2009;18:3353–64.
Mizuno Y, Amari M, Takatama M, Aizawa H, Mihara B, Okamoto K. Immunoreactivities of p62, an ubiqutin-binding protein, in the spinal anterior horn cells of patients with amyotrophic lateral sclerosis. J Neurol Sci. Elsevier. 2006;249:13–8.
Fecto F, Yan J, Vemula SP, Liu E, Yang Y, Chen W, et al. SQSTM1 mutations in familial and sporadic amyotrophic lateral sclerosis. Arch Neurol. American Medical Association. 2011;68:1440–6.
Tanji K, Zhang H-X, Mori F, Kakita A, Takahashi H, Wakabayashi K. p62/sequestosome 1 binds to TDP-43 in brains with frontotemporal lobar degeneration with TDP-43 inclusions. J Neurosci Res. John Wiley & Sons, Ltd. 2012;90:2034–42.
Nakano T, Nakaso K, Nakashima K, Ohama E. Expression of ubiquitin-binding protein p62 in ubiquitin-immunoreactive intraneuronal inclusions in amyotrophic lateral sclerosis with dementia: analysis of five autopsy cases with broad clinicopathological spectrum. Acta Neuropathol. Springer. 2004;107:359–64.
Colombrita C, Zennaro E, Fallini C, Weber M, Sommacal A, Buratti E, et al. TDP-43 is recruited to stress granules in conditions of oxidative insult. J Neurochem. John Wiley & Sons, Ltd (10.1111). 2009;111:1051–61.
Liu-Yesucevitz L, Bilgutay A, Zhang YJ, Vanderwyde T, Citro A, Mehta T, et al. Tar DNA binding protein-43 (TDP-43) associates with stress granules: analysis of cultured cells and pathological brain tissue. PLoS One. 2010;5:e13250.
Volkening K, Leystra-Lantz C, Yang W, Jaffee H, Strong MJ. Tar DNA binding protein of 43 kDa (TDP-43), 14-3-3 proteins and copper/zinc superoxide dismutase (SOD1) interact to modulate NFL mRNA stability. Implications for altered RNA processing in amyotrophic lateral sclerosis (ALS). Brain Res. Elsevier B.V. 2009;1305:168–82.
Dormann D, Rodde R, Edbauer D, Bentmann E, Fischer I, Hruscha A, et al. ALS-associated fused in sarcoma (FUS) mutations disrupt Transportin-mediated nuclear import. EMBO J. John Wiley & Sons, Ltd. 2010;29:2841–57.
McGurk L, Lee VM, Trojanowksi JQ, Van Deerlin VM, Lee EB, Bonini NM. Poly-a binding Protein-1 localization to a subset of TDP-43 inclusions in amyotrophic lateral sclerosis occurs more frequently in patients harboring an expansion in C9orf72. J Neuropathol Exp Neurol. Lippincott Williams and Wilkins. 2014;73:837–45.
Bentmann E, Neumann M, Tahirovic S, Rodde R, Dormann D, Haass C. Requirements for stress granule recruitment of fused in sarcoma (FUS) and TAR DNA-binding protein of 43 kDa (TDP-43). J Biol Chem. American Society for Biochemistry and Molecular Biology. 2012;287:23079–94.
Wolozin B, Ivanov P. Stress granules and neurodegeneration. Nat Rev Neurosci. Nature Publishing Group. 2019;20:649–66.
Protter DSW, Parker R. Principles and properties of stress granules. Trends Cell Biol. Elsevier. 2016;26:668–79.
Boeynaems S, Alberti S, Fawzi NL, Mittag T, Polymenidou M, Rousseau F, et al. Protein phase separation: a new phase in cell biology. Trends Cell Biol. Elsevier Ltd. 2018;28:420–35.
Kedersha NL, Gupta M, Li W, Miller I, Anderson P. RNA-binding proteins TIA-1 and TIAR link the phosphorylation of eIF-2α to the assembly of mammalian stress granules. J Cell Biol. The Rockefeller University Press. 1999;147:1431–41.
Kedersha N, Cho MR, Li W, Yacono PW, Chen S, Gilks N, et al. Dynamic shuttling of TIA-1 accompanies the recruitment of mRNA to mammalian stress granules. J Cell Biol. The Rockefeller University Press. 2000;151:1257–68.
Tourrière H, Chebli K, Zekri L, Courselaud B, Blanchard JM, Bertrand E, et al. The RasGAP-associated endoribonuclease G3BP assembles stress granules. J Cell Biol. The Rockefeller University Press. 2003;160:823–31.
Matsuki H, Takahashi M, Higuchi M, Makokha GN, Oie M, Fujii M. Both G3BP1 and G3BP2 contribute to stress granule formation. Genes Cells. John Wiley & Sons, Ltd. 2013;18:135–46.
Mackenzie IR, Nicholson AM, Sarkar M, Messing J, Purice MD, Pottier C, et al. TIA1 Mutations in Amyotrophic Lateral Sclerosis and Frontotemporal Dementia Promote Phase Separation and Alter Stress Granule Dynamics. Neuron. Cell Press. 2017;95:808–816.e9.
McDonald KK, Aulas A, Destroismaisons L, Pickles S, Beleac E, Camu W, et al. TAR DNA-binding protein 43 (TDP-43) regulates stress granule dynamics via differential regulation of G3BP and TIA-1. Hum Mol Genet. Oxford University Press. 2011;20:1400–10.
Khalfallah Y, Kuta R, Grasmuck C, Prat A, Durham HD, Vande Velde C. TDP-43 regulation of stress granule dynamics in neurodegenerative disease-relevant cell types. Sci Rep. Nature Publishing Group. 2018;8:7551.
Dewey CM, Cenik B, Sephton CF, Dries DR, Mayer P, Good SK, et al. TDP-43 is directed to stress granules by sorbitol, a novel physiological osmotic and oxidative stressor. Mol Cell Biol. American Society for Microbiology. 2011;31:1098–108.
Chen Y, Cohen TJ. Aggregation of the nucleic acid– binding protein TDP-43 occurs via distinct routes that are coordinated with stress granule formation. J Biol Chem. American Society for Biochemistry and Molecular Biology Inc. 2019;294:jbc.RA118.006351.
Zhang P, Fan B, Yang P, Temirov J, Messing J, Kim HJ, et al. Chronic optogenetic induction of stress granules is cytotoxic and reveals the evolution of ALS-FTD pathology. Elife. 2019;8:e39578.
Gasset-Rosa F, Lu S, Yu H, Chen C, Melamed Z, Guo L, et al. Cytoplasmic TDP-43 De-mixing Independent of Stress Granules Drives Inhibition of Nuclear Import, Loss of Nuclear TDP-43, and Cell Death. Neuron. Elsevier. 2019;102:339–357.e7.
Babinchak WM, Haider R, Dumm BK, Sarkar P, Surewicz K, Choi JK, et al. The role of liquid-liquid phase separation in aggregation of the TDP-43 low-complexity domain. J Biol Chem. 2019;294:6306–17.
Mann JR, Gleixner AM, Mauna JC, Gomes E, DeChellis-Marks MR, Needham PG, et al. RNA Binding Antagonizes Neurotoxic Phase Transitions of TDP-43. Neuron. Elsevier. 2019;102:321–338.e8.
McGurk L, Gomes E, Guo L, Mojsilovic-Petrovic J, Tran V, Kalb RG, et al. Poly(ADP-ribose) prevents pathological phase separation of TDP-43 by promoting liquid Demixing and stress granule localization. Mol Cell. 2018;71:703–717.e9.
Scotter EL, Vance C, Nishimura AL, Lee Y-BYB, Chen HJH-J, Urwin H, et al. Differential roles of the ubiquitin proteasome system and autophagy in the clearance of soluble and aggregated TDP-43 species. J Cell Sci. Company of Biologists. 2014;127:1263–78.
Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N, et al. Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature. Nature Publishing Group. 2011;477:211–5.
Picher-Martel V, Renaud L, Bareil C, Julien JP. Neuronal expression of UBQLN2P497H exacerbates TDP-43 pathology in TDP-43G348C mice through interaction with ubiquitin. Mol Neurobiol. 2018;56:1–17.
Le NTT, Chang L, Kovlyagina I, Georgiou P, Safren N, Braunstein KE, et al. Motor neuron disease, TDP-43 pathology, and memory deficits in mice expressing ALS-FTD-linked UBQLN2 mutations. Proc Natl Acad Sci U S A. National Academy of Sciences. 2016;113:E7580–9.
Wu Q, Liu M, Huang C, Liu X, Huang B, Li N, et al. Pathogenic Ubqln2 gains toxic properties to induce neuron death. Acta Neuropathol. Springer Verlag. 2015;129:417–28.
Millecamps S, Corcia P, Cazeneuve C, Boillée S, Seilhean D, Danel-Brunaud V, et al. Mutations in UBQLN2 are rare in French amyotrophic lateral sclerosis. Neurobiol Aging. Elsevier Inc. 2012;33:839.e1–3.
van Doormaal PTC, van Rheenen W, van Blitterswijk M, Schellevis RD, Schelhaas HJ, de Visser M, et al. UBQLN2 in familial amyotrophic lateral sclerosis in the Netherlands. Neurobiol Aging. Elsevier Inc. 2012;33:2233.e7–8.
Daoud H, Suhail H, Szuto A, Camu W, Salachas F, Meininger V, et al. UBQLN2 mutations are rare in French and French-Canadian amyotrophic lateral sclerosis. Neurobiol Aging. Elsevier Inc. 2012;33:2230.e1–5.
van Eersel J, Ke YD, Gladbach A, Bi M, Götz J, Kril JJ, et al. Cytoplasmic accumulation and aggregation of TDP-43 upon proteasome inhibition in cultured neurons. Iijima KM, editor. PLoS One. Public Library of Science. 2011;6:e22850.
Ishii T, Kawakami E, Endo K, Misawa H, Watabe K. Formation and spreading of TDP-43 aggregates in cultured neuronal and glial cells demonstrated by time-lapse imaging. PLoS One. Public Library of Science. 2017;12:e0179375.
Dobson-Stone C, Hallupp M, Shahheydari H, Ragagnin AMG, Chatterton Z, Carew-Jones F, et al. CYLD is a causative gene for frontotemporal dementia-amyotrophic lateral sclerosis. Brain. 2020;143:783–99.
Maruyama H, Morino H, Ito H, Izumi Y, Kato H, Watanabe Y, et al. Mutations of optineurin in amyotrophic lateral sclerosis. Nature. Nature Publishing Group. 2010;465:223–6.
Freischmidt A, Wieland T, Richter B, Ruf W, Schaeffer V, Müller K, et al. Haploinsufficiency of TBK1 causes familial ALS and fronto-temporal dementia. Nat Neurosci. Nature Publishing Group. 2015;18:631–6.
de Majo M, Topp SD, Smith BN, Nishimura AL, Chen HJ, Gkazi AS, et al. ALS-associated missense and nonsense TBK1 mutations can both cause loss of kinase function. Neurobiol Aging. Elsevier Inc. 2018;71:266.e1–266.e10.
Weinreich M, Shepheard SR, Verber N, Wyles M, Heath PR, Highley JR, et al. Neuropathological characterization of a novel TANK binding kinase (TBK1) gene loss of function mutation associated with amyotrophic lateral sclerosis. Neuropathol Appl Neurobiol. 2019;46:279–91.
Brady OA, Meng P, Zheng Y, Mao Y, Hu F. Regulation of TDP-43 aggregation by phosphorylation andp62/SQSTM1. J Neurochem. 2011;116:248–59.
Gal J, Ström AL, Kilty R, Zhang F, Zhu H. p62 accumulates and enhances aggregate formation in model systems of familial amyotrophic lateral sclerosis. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2007;282:11068–77.
Seibenhener ML, Babu JR, Geetha T, Wong HC, Krishna NR, Wooten MW. Sequestosome 1/p62 is a Polyubiquitin chain binding protein involved in ubiquitin proteasome degradation. Mol Cell Biol. American Society for Microbiology. 2004;24:8055–68.
Fallini C, Bassell GJ, Rossoll W. The ALS disease protein TDP-43 is actively transported in motor neuron axons and regulates axon outgrowth. Hum Mol Genet. 2012;21:3703–18.
Alami NH, Smith RB, Carrasco MA, Williams LA, Winborn CS, Han SSW, et al. Axonal transport of TDP-43 mRNA granules is impaired by ALS-causing mutations. Neuron. 2014;81:536–43.
Nicolas A, Kenna KP, Renton AE, Shaw CE, Traynor BJ, Landers Correspondence JE, et al. Genome-wide Analyses Identify KIF5A as a Novel ALS Gene. Neuron. 2018;97:1268–1282.e6.
Brenner D, Yilmaz R, Müller K, Grehl T, Petri S, Meyer T, et al. Hot-spot KIF5A mutations cause familial ALS. Brain. 2018;141:688–97.
Puls I, Jonnakuty C, LaMonte BH, Holzbaur ELF, Tokito M, Mann E, et al. Mutant dynactin in motor neuron disease. Nat Genet. Nature Publishing Group. 2003;33:455–6.
Münch C, Sedlmeier R, Meyer T, Homberg V, Sperfeld AD, Kurt A, et al. Point mutations of the p150 subunit of dynactin (DCTN1) gene in ALS. Neurology. 2004;63:724–6.
Kieran D, Hafezparast M, Bohnert S, Dick JRT, Martin J, Schiavo G, et al. A mutation in dynein rescues axonal transport defects and extends the life span of ALS mice. J Cell Biol. The Rockefeller University Press. 2005;169:561–7.
Bilsland LG, Sahai E, Kelly G, Golding M, Greensmith L, Schiavo G. Deficits in axonal transport precede ALS symptoms in vivo. Proc Natl Acad Sci U S A. National Academy of Sciences. 2010;107:20523–8.
De Vos KJ, Hafezparast M. Neurobiology of axonal transport defects in motor neuron diseases: Opportunities for translational research? Neurobiol Dis. Academic Press Inc. 2017;105:283–99.
Sleigh JN, Tosolini AP, Gordon D, Devoy A, Fratta P, Fisher EMC, et al. ALS mice carrying pathological mutant TDP-43, but not mutant FUS, display axonal transport defects in vivo. Cell Rep. 2020;31:3655–62.
Coyne AN, Siddegowda BB, Estes PS, Johannesmeyer J, Kovalik T, Daniel SG, et al. FUTSCH/MAP1B mRNA is a translational target of TDP-43 and is neuroprotective in a Drosophila model of amyotrophic lateral sclerosis. J Neurosci. Society for Neuroscience. 2014;34:15962–74.
Majumder P, Chu JF, Chatterjee B, Swamy KBS, Shen CKJ. Co-regulation of mRNA translation by TDP-43 and fragile X syndrome protein FMRP. Acta Neuropathol. Springer Verlag. 2016;132:721–38.
Arrasate M, Mitra S, Schweitzer ES, Segal MR, Finkbeiner S. Inclusion body formation reduces levels of mutant huntingtin and the risk of neuronal death. Nature. Nature Publishing Group. 2004;431:805–10.
Parone PA, Da Cruz S, Han JS, McAlonis-Downes M, Vetto AP, Lee SK, et al. Enhancing mitochondrial calcium buffering capacity reduces aggregation of misfolded SOD1 and motor neuron cell death without extending survival in mouse models of inherited amyotrophic lateral sclerosis. J Neurosci. Society for Neuroscience. 2013;33:4657–71.
Treusch S, Cyr DM, Lindquist S. Amyloid deposits: Protection against toxic protein species? Cell Cycle. Taylor and Francis Inc. 2009;8:1668–74.
Kitamura A, Yuno S, Muto H, Kinjo M. Different aggregation states of a nuclear localization signal-tagged 25-kDa C-terminal fragment of TAR RNA/DNA-binding protein 43 kDa. Genes Cells. Blackwell Publishing Ltd. 2017;22:521–34.
French RL, Grese ZR, Aligireddy H, Dhavale DD, Reeb AN, Kedia N, et al. Detection of TAR DNA-binding protein 43 (TDP-43) oligomers as initial intermediate species during aggregate formation. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2019;294:6696–709.
Fang YS, Tsai KJ, Chang YJ, Kao P, Woods R, Kuo PH, et al. Full-length TDP-43 forms toxic amyloid oligomers that are present in frontotemporal lobar dementia-TDP patients. Nat Commun. Nature Publishing Group. 2014;5:1–13.
Cao Q, Boyer DR, Sawaya MR, Ge P, Eisenberg DS. Cryo-EM structures of four polymorphic TDP-43 amyloid cores. Nat Struct Mol Biol. Nature Publishing Group. 2019;26:619–27.
Laferrière F, Maniecka Z, Pérez-Berlanga M, Hruska-Plochan M, Gilhespy L, Hock E-M, et al. TDP-43 extracted from frontotemporal lobar degeneration subject brains displays distinct aggregate assemblies and neurotoxic effects reflecting disease progression rates. Nat Neurosci. Nature Publishing Group. 2019;22:65–77.
Geser F, Robinson JL, Malunda JA, Xie SX, Clark CM, Kwong LK, et al. Pathological 43-kDa transactivation response DNA-binding protein in older adults with and without severe mental illness. Arch Neurol. NIH Public Access. 2010;67:1238–50.
Liu R, Yang G, Nonaka T, Arai T, Jia W, Cynader MS. Reducing TDP-43 aggregation does not prevent its cytotoxicity. Acta Neuropathol Commun. BioMed Central. 2014;2:49.
Barmada SJ, Skibinski G, Korb E, Rao EJ, Wu JY, Finkbeiner S. Cytoplasmic mislocalization of TDP-43 is toxic to neurons and enhanced by a mutation associated with familial amyotrophic lateral sclerosis. J Neurosci. 2010;30:639–49.
Igaz LM, Kwong LK, Lee EB, Chen-Plotkin A, Swanson E, Unger T, et al. Dysregulation of the ALS-associated gene TDP-43 leads to neuronal death and degeneration in mice. J Clin Invest. American Society for Clinical Investigation. 2011;121:726–38.
Walker AK, Spiller KJ, Ge G, Zheng A, Xu Y, Zhou M, et al. Functional recovery in new mouse models of ALS/FTLD after clearance of pathological cytoplasmic TDP-43. Acta Neuropathol. Springer Berlin Heidelberg. 2015;130:643–60.
Sasaguri H, Chew J, Xu YF, Gendron TF, Garrett A, Lee CW, et al. The extreme N-terminus of TDP-43 mediates the cytoplasmic aggregation of TDP-43 and associated toxicity in vivo. Brain Res. Elsevier BV. 2016;1647:57–64.
Arnold ES, Ling S-C, Huelga SC, Lagier-Tourenne C, Polymenidou M, Ditsworth D, et al. ALS-linked TDP-43 mutations produce aberrant RNA splicing and adult-onset motor neuron disease without aggregation or loss of nuclear TDP-43. Proc Natl Acad Sci U S A. National Academy of Sciences. 2013;110:E736–45.
Maniecka Z, Polymenidou M. From nucleation to widespread propagation: a prion-like concept for ALS. Virus Res. Elsevier. 2015;207:94–105.
Porta S, Xu Y, Restrepo CR, Kwong LK, Zhang B, Brown HJ, et al. Patient-derived frontotemporal lobar degeneration brain extracts induce formation and spreading of TDP-43 pathology in vivo. Nat Commun. Nature Publishing Group. 2018;9:4220.
Mori F, Tada M, Kon T, Miki Y, Tanji K, Kurotaki H, et al. Phosphorylated TDP-43 aggregates in skeletal and cardiac muscle are a marker of myogenic degeneration in amyotrophic lateral sclerosis and various conditions. Acta Neuropathol Commun. BioMed Central Ltd. 2019;7:165.
Vogler TO, Wheeler JR, Nguyen ED, Hughes MP, Britson KA, Lester E, et al. TDP-43 and RNA form amyloid-like myo-granules in regenerating muscle. Nature. Nature Publishing Group. 2018;563:508–13.
Cykowski MD, Powell SZ, Appel JW, Arumanayagam AS, Rivera AL, Appel SH. Phosphorylated TDP-43 (pTDP-43) aggregates in the axial skeletal muscle of patients with sporadic and familial amyotrophic lateral sclerosis. Acta Neuropathol Commun. NLM (Medline). 2018;6:28.
Guerrero EN, Mitra J, Wang H, Rangaswamy S, Hegde PM, Basu P, et al. Amyotrophic lateral sclerosis (ALS)-associated TDP-43 mutation Q331K prevents nuclear translocation of XRCC4-DNA ligase 4 complex and is linked to genome damage-mediated neuronal apoptosis. Hum Mol Genet. 2019;28(5):2459–76.
D’Amico E, Factor-Litvak P, Santella RM, Mitsumoto H. Clinical perspective on oxidative stress in sporadic amyotrophic lateral sclerosis. Free Radic Biol Med. NIH Public Access. 2013;65:509–27.
Asakawa K, Handa H, Kawakami K. Optogenetic modulation of TDP-43 oligomerization accelerates ALS-related pathologies in the spinal motor neurons. Nat Commun. Nature Research. 2020;11:1–16.
Modic M, Rot G, Grosch M, Lepko T, Shaposhnikov D, Cacchiarelli D, et al. Cross-regulation between TDP-43 and Paraspeckles promotes pluripotency-differentiation transition. SSRN Electron J. 2018;74:951–65.
Melamed Z, López-Erauskin J, Baughn MW, Zhang O, Drenner K, Sun Y, et al. Premature polyadenylation-mediated loss of stathmin-2 is a hallmark of TDP-43-dependent neurodegeneration. Nat Neurosci. 2019;22:180–90.
Ling JP, Pletnikova O, Troncoso JC, Wong PC. TDP-43 repression of nonconserved cryptic exons is compromised in ALS-FTD. Science. American Association for the Advancement of Science. 2015;349:650–5.
Buratti E. Multiple roles of TDP-43 in gene expression, splicing regulation, and human disease. Front Biosci. 2008;13:867–78.
Tollervey JR, Curk T, Rogelj B, Briese M, Cereda M, Kayikci M, et al. Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat Neurosci. 2011;14:452–8.
Buratti E, Dörk T, Zuccato E, Pagani F, Romano M, Baralle FE. Nuclear factor TDP-43 and SR proteins promote in vitro and in vivo CFTR exon 9 skipping. EMBO J. 2001;20:1774–84.
Rot G, Wang Z, Huppertz I, Modic M, Lenče T, Hallegger M, et al. High-resolution RNA maps suggest common principles of splicing and polyadenylation regulation by TDP-43. Cell Rep. Elsevier B.V. 2017;19:1056–67.
Polymenidou M, Lagier-Tourenne C, Hutt KR, Huelga SC, Moran J, Liang TY, et al. Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat Neurosci. Nat Publ Group. 2011;14:459–68.
Klim JR, Williams LA, Limone F, Guerra San Juan I, Davis-Dusenbery BN, Mordes DA, et al. ALS-implicated protein TDP-43 sustains levels of STMN2, a mediator of motor neuron growth and repair. Nat Neurosci. Nature Publishing Group. 2019;22:167–79.
Chu JF, Majumder P, Chatterjee B, Huang SL, Shen CKJ. TDP-43 Regulates Coupled Dendritic mRNA Transport-Translation Processes in Co-operation with FMRP and Staufen1. Cell Rep. Elsevier B.V. 2019;29:3118–3133.e6.
Amlie-Wolf A, Ryvkin P, Tong R, Dragomir I, Suh E, Xu Y, et al. Transcriptomic changes due to cytoplasmic TDP-43 expression reveal dysregulation of histone transcripts and nuclear chromatin. Buratti E, editor. PLoS One. 2015;10:e0141836.
Wu LS, Cheng WC, Chen CY, Wu MC, Wang YC, Tseng YH, et al. Transcriptomopathies of pre- and post-symptomatic frontotemporal dementia-like mice with TDP-43 depletion in forebrain neurons. Acta Neuropathol Commun. NLM (Medline). 2019;7:50.
Jeong YH, Ling JP, Lin SZ, Donde AN, Braunstein KE, Majounie E, et al. Tdp-43 cryptic exons are highly variable between cell types. Mol Neurodegener. 2017;12:13.
Ishiguro A, Kimura N, Noma T, Shimo-Kon R, Ishihama A, Kon T. Molecular dissection of ALS-linked TDP-43: involvement of the Gly-rich domain in interaction with G-quadruplex mRNA. FEBS Lett. 2020. https://doi.org/10.1002/1873-3468.13800.
Ishiguro A, Kimura N, Watanabe Y, Watanabe S, Ishihama A. TDP-43 binds and transports G-quadruplex-containing mRNAs into neurites for local translation. Genes to Cells. Blackwell Publishing Ltd. 2016;21:466–81.
Zhou B, Liu C, Geng Y, Zhu G. Topology of a G-quadruplex DNA formed by C9orf72 hexanucleotide repeats associated with ALS and FTD. Sci Rep. Nature Publishing Group. 2015;5:1–7.
Conlon EG, Lu L, Sharma A, Yamazaki T, Tang T, Shneider NA, et al. The C9ORF72 GGGGCC expansion forms RNA G-quadruplex inclusions and sequesters hnRNP H to disrupt splicing in ALS brains. Elife. eLife Sciences Publications Ltd. 2016;5:e17820.
Fay MM, Anderson PJ, Ivanov P. ALS/FTD-associated C9ORF72 repeat RNA promotes phase transitions in vitro and in cells. Cell Rep. 2017;21:3573–84.
Elden AC, Kim HJ, Hart MP, Chen-Plotkin AS, Johnson BS, Fang X, et al. Ataxin-2 intermediate-length polyglutamine expansions are associated with increased risk for ALS. Nature. Nature Publishing Group. 2010;466:1069–75.
Flores BN, Li X, Martinez J, Beg AA, Barmada SJ. An Intramolecular Salt Bridge Linking TDP43’s RNA Recognition Motifs Dictates RNA Binding and TDP43-Dependent Neurodegeneration. Cell Rep. Cell Press. 2019;27:1133–1150.e8.
Ihara R, Matsukawa K, Nagata Y, Kunugi H, Tsuji S, Chihara T, et al. RNA binding mediates neurotoxicity in the transgenic Drosophila model of TDP-43 proteinopathy. Hum Mol Genet. 2013;22:4474–84.
Voigt A, Herholz D, Fiesel FC, Kaur K, Müller D, Karsten P, et al. TDP-43-mediated neuron loss in vivo requires RNA-binding activity. Feany MB, editor. PLoS One. Public Library of Science. 2010;5:e12247.
Chen H-J, Topp SD, Hui HS, Zacco E, Katarya M, McLoughlin C, et al. RRM adjacent TARDBP mutations disrupt RNA binding and enhance TDP-43 proteinopathy. Brain. 2019;142:3753–70.
Maharana S, Wang J, Papadopoulos DK, Richter D, Pozniakovsky A, Poser I, et al. RNA buffers the phase separation behavior of prion-like RNA binding proteins. Science. American Association for the Advancement of Science. 2018;360:918–21.
Lalmansingh AS, Urekar CJ, Reddi PP. TDP-43 is a transcriptional repressor: the testis-specific mouse acrv1 gene is a TDP-43 target in vivo. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2011;286:10970–82.
Schwenk BM, Hartmann H, Serdaroglu A, Schludi MH, Hornburg D, Meissner F, et al. TDP-43 loss of function inhibits endosomal trafficking and alters trophic signaling in neurons. EMBO J. John Wiley & Sons, Ltd. 2016;35:2350–70.
Cox LE, Ferraiuolo L, Goodall EF, Heath PR, Higginbottom A, Mortiboys H, et al. Mutations in CHMP2B in lower motor neuron predominant amyotrophic lateral sclerosis (ALS). PLoS One. 2010;5:e9872.
Takahashi Y, Fukuda Y, Yoshimura J, Toyoda A, Kurppa K, Moritoyo H, et al. Erbb4 mutations that disrupt the neuregulin-erbb4 pathway cause amyotrophic lateral sclerosis type 19. Am J Hum Genet. Cell Press. 2013;93:900–5.
Kim BW, Jeong YE, Wong M, Martin LJ. DNA damage accumulates and responses are engaged in human ALS brain and spinal motor neurons and DNA repair is activatable in iPSC-derived motor neurons with SOD1 mutations. Acta Neuropathol Commun. BioMed Central Ltd. 2020;8:1–26.
Mitra J, Guerrero EN, Hegde PM, Liachko NF, Wang H, Vasquez V, et al. Motor neuron disease-associated loss of nuclear TDP-43 is linked to DNA double-strand break repair defects. Proc Natl Acad Sci. National Academy of Sciences. 2019;116:4696–705.
Shaikh AY, Martin LJ. DNA base-excision repair enzyme apurinic/apyrimidinic endonuclease/redox factor-1 is increased and competent in the brain and spinal cord of individuals with amyotrophic lateral sclerosis. NeuroMolecular Med. Springer Science and Business Media LLC. 2002;2:47–60.
Kim SH, Engelhardt JI, Henkel JS, Siklós L, Soós J, Goodman C, et al. Widespread increased expression of the DNA repair enzyme PARP in brain in ALS. Neurology. 2004;62:319–22.
Olkowski ZL. Mutant AP endonuclease in patients with amyotrophic lateral sclerosis. Neuroreport. Lippincott Williams and Wilkins. 1998;9:239–42.
Higelin J, Catanese A, Semelink-Sedlacek LL, Oeztuerk S, Lutz AK, Bausinger J, et al. NEK1 loss-of-function mutation induces DNA damage accumulation in ALS patient-derived motoneurons. Stem Cell Res. Elsevier B.V. 2018;30:150–62.
Kawaguchi T, Rollins MG, Moinpour M, Morera AA, Ebmeier CC, Old WM, et al. Changes to the TDP-43 and FUS Interactomes induced by DNA damage. J Proteome Res. 2020;19:360–70.
Roos WP, Kaina B. DNA damage-induced cell death by apoptosis. Trends Mol Med. 2006;12:440–50.
Wu C, Jin L, Wang I, Wei W, Ho P, Liu Y, et al. HDAC1 dysregulation induces aberrant cell cycle and DNA damage in progress of TDP-43 proteinopathies. EMBO Mol Med. John Wiley & Sons, Ltd. 2020;12(6):e10622.
Tibshirani M, Zhao B, Gentil BJ, Minotti S, Marques C, Keith J, et al. Dysregulation of chromatin remodelling complexes in amyotrophic lateral sclerosis. Hum Mol Genet. 2017;26:4142–52.
Liu EY, Russ J, Cali CP, Phan JM, Amlie-Wolf A, Lee Correspondence EB. Loss of nuclear TDP-43 is associated with Decondensation of LINE retrotransposons. Cell Rep. 2019;27:1409–1421.e6.
Birsa N, Bentham MP, Fratta P. Cytoplasmic functions of TDP-43 and FUS and their role in ALS. Semin Cell Dev Biol. Academic Press. 2019;99:193–201.
Leibiger C, Deisel J, Aufschnaiter A, Ambros S, Tereshchenko M, Verheijen BM, et al. TDP-43 controls lysosomal pathways thereby determining its own clearance and cytotoxicity. Hum Mol Genet. 2018;27:1593–607.
Liu G, Coyne AN, Pei F, Vaughan S, Chaung M, Zarnescu DC, et al. Endocytosis regulates TDP-43 toxicity and turnover. Nat Commun. Nature Publishing Group. 2017;8:2092.
Liu G, Byrd A, Warner AN, Pei F, Basha E, Buchanan A, et al. Cdc48/VCP and Endocytosis Regulate TDP-43 and FUS Toxicity and Turnover. Mol Cell Biol. American Society for Microbiology. 2019;40:e00256–19.
Smethurst P, Risse E, Tyzack GE, Mitchell JS, Taha DM, Chen YR, et al. Distinct responses of neurons and astrocytes to TDP-43 proteinopathy in amyotrophic lateral sclerosis. Brain. 2020;143:430–40.
Kawamata H, Peixoto P, Konrad C, Palomo G, Bredvik K, Gerges M, et al. Mutant TDP-43 does not impair mitochondrial bioenergetics in vitro and in vivo. Mol Neurodegener. 2017;12:37.
Benajiba L, Le BI, Camuzat A, Lacoste M, Thomas-Anterion C, Couratier P, et al. TARDBP mutations in motoneuron disease with frontotemporal lobar degeneration. Ann Neurol. John Wiley and Sons Inc. 2009;65:470–3.
Borghero G, Pugliatti M, Marrosu F, Marrosu MG, Murru MR, Floris G, et al. Genetic architecture of ALS in Sardinia. Neurobiol Aging. Elsevier Inc. 2014;35:2882.e7–2882.e12.
Millecamps S, Salachas F, Cazeneuve C, Gordon P, Bricka B, Camuzat A, et al. SOD1, ANG, VAPB, TARDBP, and FUS mutations in familial amyotrophic lateral sclerosis: genotype-phenotype correlations. J Med Genet. BMJ Publishing Group Ltd. 2010;47:554–60.
Ticozzi N, LeClerc AL, van Blitterswijk M, Keagle P, McKenna-Yasek DM, Sapp PC, et al. Mutational analysis of TARDBP in neurodegenerative diseases. Neurobiol Aging. Elsevier. 2011;32:2096–9.
Van Blitterswijk M, Van Es MA, Hennekam EAM, Dooijes D, Van Rheenen W, Medic J, et al. Evidence for an oligogenic basis of amyotrophic lateral sclerosis. Hum Mol Genet. 2012;21:3776–84.
Nozaki I, Arai M, Takahashi K, Hamaguchi T, Yoshikawa H, Muroishi T, et al. Familial ALS with G298S mutation in TARDBP: a comparison of CSF tau protein levels with those in sporadic ALS. Intern Med. The Japanese Society of Internal Medicine. 2010;49:1209–12.
Winton MJ, Van Deerlin VM, Kwong LK, Yuan W, Wood EMC, Yu CE, et al. A90V TDP-43 variant results in the aberrant localization of TDP-43 in vitro. FEBS Lett. NIH Public Access. 2008s;582:2252–6.
Conforti FL, Sproviero W, Simone IL, Mazzei R, Valentino P, Ungaro C, et al. TARDBP gene mutations in south Italian patients with amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry. BMJ Publishing Group Ltd. 2011;82:587–8.
Corrado L, Ratti A, Gellera C, Buratti E, Castellotti B, Carlomagno Y, et al. High frequency of TARDBP gene mutations in italian patients with amyotrophic lateral sclerosis. Hum Mutat. Wiley-Liss Inc. 2009;30:688–94.
Del Bo R, Ghezzi S, Corti S, Pandolfo M, Ranieri M, Santoro D, et al. TARDBP (TDP-43) sequence analysis in patients with familial and sporadic ALS: identification of two novel mutations. Eur J Neurol. John Wiley & Sons, Ltd. 2009;16:727–32.
Williams KL, Durnall JC, Thoeng AD, Warraich ST, Nicholson GA, Blair IP. A novel TARDBP mutation in an Australian amyotrophic lateral sclerosis kindred. J Neurol Neurosurg Psychiatry. BMJ Publishing Group. 2009;80:1286–8.
Pamphlett R, Luquin N, McLean C, Jew SK, Adams L. TDP-43 neuropathology is similar in sporadic amyotrophic lateral sclerosis with or without TDP-43 mutations. Neuropathol Appl Neurobiol. John Wiley & Sons, Ltd. 2009;35:222–5.
Aizawa H, Yamashita T, Kato H, Kimura T, Kwak S. Impaired nucleoporins are present in sporadic amyotrophic lateral sclerosis motor neurons that exhibit mislocalization of the 43-kDa TAR DNA-binding protein. J Clin Neurol. Korean Neurological Association. 2019;15:62–7.
Chou CC, Zhang YJY, Umoh ME, Vaughan SW, Lorenzini I, Liu F, et al. TDP-43 pathology disrupts nuclear pore complexes and nucleocytoplasmic transport in ALS/FTD. Nat Neurosci. Nature Publishing Group. 2018;21:228–39.
Zhang K, Donnelly CJ, Haeusler AR, Grima JC, Machamer JB, Steinwald P, et al. The C9orf72 repeat expansion disrupts nucleocytoplasmic transport. Nature. Nature Publishing Group. 2015;525:56–61.
Renton AE, Majounie E, Waite A, Simón-Sánchez J, Rollinson S, Gibbs JR, et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72:257–68.
Majounie E, Renton AE, Mok K, Dopper EGP, Waite A, Rollinson S, et al. Frequency of the C9orf72 hexanucleotide repeat expansion in patients with amyotrophic lateral sclerosis and frontotemporal dementia: a cross-sectional study. Lancet Neurol. Lancet Publishing Group. 2012;11:323–30.
Ditsworth D, Maldonado M, McAlonis-Downes M, Sun S, Seelman A, Drenner K, et al. Mutant TDP-43 within motor neurons drives disease onset but not progression in amyotrophic lateral sclerosis. Acta Neuropathol. Springer Verlag. 2017;133:907–22.
Boeynaems S, Bogaert E, Michiels E, Gijselinck I, Sieben A, Jovičić A, et al. Drosophila screen connects nuclear transport genes to DPR pathology in c9ALS/FTD. Sci Rep. Nature Publishing Group. 2016;6:20877.
Freibaum BD, Lu Y, Lopez-Gonzalez R, Kim NC, Almeida S, Lee KH, et al. GGGGCC repeat expansion in C9orf72 compromises nucleocytoplasmic transport. Nature. Nature Publishing Group. 2015;525:129–33.
Jovičič A, Mertens J, Boeynaems S, Bogaert E, Chai N, Yamada SB, et al. Modifiers of C9orf72 dipeptide repeat toxicity connect nucleocytoplasmic transport defects to FTD/ALS. Nat Neurosci. Nature Publishing Group. 2015;18:1226–9.
Shi KY, Mori E, Nizami ZF, Lin Y, Kato M, Xiang S, et al. Toxic PRn poly-dipeptides encoded by the C9orf72 repeat expansion block nuclear import and export. Proc Natl Acad Sci U S A. National Academy of Sciences. 2017;114:E1111–7.
Zu T, Liu Y, Bañez-Coronel M, Reid T, Pletnikova O, Lewis J, et al. RAN proteins and RNA foci from antisense transcripts in C9ORF72 ALS and frontotemporal dementia. Proc Natl Acad Sci U S A. 2013;110:E4968–77.
Mori K, Arzberger T, Grässer FA, Gijselinck I, May S, Rentzsch K, et al. Bidirectional transcripts of the expanded C9orf72 hexanucleotide repeat are translated into aggregating dipeptide repeat proteins. Acta Neuropathol. 2013;126:881–93.
Solomon DA, Stepto A, Au WH, Adachi Y, Diaper DC, Hall R, et al. A feedback loop between dipeptide-repeat protein, TDP-43 and karyopherin-α mediates C9orf72-related neurodegeneration. Brain. 2018;141:2908–24.
Buratti E. TDP-43 post-translational modifications in health and disease. Expert Opin Ther Targets. 2018;22(3):279–93.
Liachko NF, McMillan PJ, Guthrie CR, Bird TD, Leverenz JB, Kraemer BC. CDC7 inhibition blocks pathological TDP-43 phosphorylation and neurodegeneration. Ann Neurol. John Wiley & Sons, Ltd. 2013;74:39–52.
Alquezar C, Salado IG, De La Encarnación A, Pérez DI, Moreno F, Gil C, et al. Targeting TDP-43 phosphorylation by casein kinase-1δ inhibitors: a novel strategy for the treatment of frontotemporal dementia. Mol Neurodegener. 2016;11:36.
Kametani F, Nonaka T, Suzuki T, Arai T, Dohmae N, Akiyama H, et al. Identification of casein kinase-1 phosphorylation sites on TDP-43. Biochem Biophys Res Commun. Academic Press. 2009;382:405–9.
Nonaka T, Suzuki G, Tanaka Y, Kametani F, Hirai S, Okado H, et al. Phosphorylation of TAR DNA-binding protein of 43 kDa (TDP-43) by truncated casein kinase 1δ triggers mislocalization and accumulation of TDP-43. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2016;291:5473–83.
Krach F, Batra R, Wheeler EC, Vu AQ, Wang R, Hutt K, et al. Transcriptome–pathology correlation identifies interplay between TDP-43 and the expression of its kinase CK1E in sporadic ALS. Acta Neuropathol. Springer Berlin Heidelberg. 2018;136:405–23.
Choksi DK, Roy B, Chatterjee S, Yusuff T, Bakhoum MF, Sengupta U, et al. TDP-43 phosphorylation by casein kinase iε promotes oligomerization and enhances toxicity in vivo. Hum Mol Genet. 2014;23:1025–35.
Liachko NF, Guthrie CR, Kraemer BC. Phosphorylation promotes neurotoxicity in a Caenorhabditis elegans model of TDP-43 proteinopathy. J Neurosci. Society for Neuroscience. 2010;30:16208–19.
Liachko NF, McMillan PJ, Strovas TJ, Loomis E, Greenup L, Murrell JR, et al. The tau tubulin kinases TTBK1/2 promote accumulation of pathological TDP-43. PLoS Genet. Public Library of Science. 2014;10:e1004803.
Taylor LM, McMillan PJ, Liachko NF, Strovas TJ, Ghetti B, Bird TD, et al. Pathological phosphorylation of tau and TDP-43 by TTBK1 and TTBK2 drives neurodegeneration. Mol Neurodegener. BioMed Central Ltd. 2018;13:7.
Li HY, Yeh PA, Chiu HC, Tang CY, Tu BP. Hyperphosphorylation as a defense mechanism to reduce TDP-43 aggregation. Ginsberg SD, editor. PLoS One. Public Library of Science. 2011;6:e23075.
Li W, Reeb AN, Lin B, Subramanian P, Fey EE, Knoverek CR, et al. Heat shock-induced phosphorylation of TAR DNA-binding protein 43 (TDP-43) by MAPK/ERK kinase regulates TDP-43 function. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2017;292:5089–100.
Ikeda K, Tsuchiya K. Motor neuron disease group accompanied by inclusions of unidentified protein signaled by ubiquitin. Neuropathology. 2004;24:117–24.
Cohen TJ, Hwang AW, Unger T, Trojanowski JQ, Lee VMY. Redox signalling directly regulates TDP-43 via cysteine oxidation and disulphide cross-linking. EMBO J. 2012;31:1241–52.
Cohen TJ, Hwang AW, Restrepo CR, Yuan CX, Trojanowski JQ, Lee VMY. An acetylation switch controls TDP-43 function and aggregation propensity. Nat Commun. Nature Publishing Group. 2015;6:5845.
Nishimoto Y, Ito D, Yagi T, Nihei Y, Tsunodo Y, Suzuki N. Characterization of alternative isoforms and inclusion body of the TAR DNA-binding protein-43. J Biol Chem. American Society for Biochemistry and Molecular Biology. 2010;285:608–19.
Salvatori I, Ferri A, Scaricamazza S, Giovannelli I, Serrano A, Rossi S, et al. Differential toxicity of TAR DNA-binding protein 43 isoforms depends on their submitochondrial localization in neuronal cells. J Neurochem. Blackwell Publishing Ltd. 2018;146:585–97.
Fung G, Shi J, Deng H, Hou J, Wang C, Hong A, et al. Cytoplasmic translocation, aggregation, and cleavage of TDP-43 by enteroviral proteases modulate viral pathogenesis. Cell Death Differ. 2015;22:2087–97.
Fratta P, Sivakumar P, Humphrey J, Lo K, Ricketts T, Oliveira H, et al. Mice with endogenous TDP-43 mutations exhibit gain of splicing function and characteristics of amyotrophic lateral sclerosis. EMBO J. 2018;37:e98684.
Weskamp K, Tank EM, Miguez R, McBride JP, Gómez NB, White M, et al. Shortened TDP43 isoforms upregulated by neuronal hyperactivity drive TDP43 pathology in ALS. J Clin Invest. 2020;130:1139–55.
Hideyama T, Teramoto S, Hachiga K, Yamashita T, Kwak S. Co-occurrence of TDP-43 Mislocalization with reduced activity of an RNA editing enzyme, ADAR2, in aged mouse motor neurons. Kano MR, editor. PLoS One. Public Library of Science. 2012;7:e43469.
De Giorgio F, Maduro C, EMC F, Acevedo-Arozena A. Transgenic and physiological mouse models give insights into different aspects of amyotrophic lateral sclerosis. Dis Model Mech. Company of Biologists Ltd. 2019;12(1):dmm037424.
Bennett CL, Dastidar SG, Ling SC, Malik B, Ashe T, Wadhwa M, et al. Senataxin mutations elicit motor neuron degeneration phenotypes and yield TDP-43 mislocalization in ALS4 mice and human patients. Acta Neuropathol. Springer Verlag. 2018;136:425–43.
Grunseich C, Patankar A, Amaya J, Watts JA, Li D, Ramirez P, et al. Clinical and molecular aspects of Senataxin mutations in amyotrophic lateral sclerosis 4. Ann Neurol. John Wiley and Sons Inc. 2020;87:547–55.
Chen YZ, Bennett CL, Huynh HM, Blair IP, Puls I, Irobi J, et al. DNA/RNA helicase gene mutations in a form of juvenile amyotrophic lateral sclerosis (ALS4). Am J Hum Genet. University of Chicago Press. 2004;74:1128–35.
Huang SL, Wu LS, Lee M, Chang CW, Cheng WC, Fang YS, et al. A robust TDP-43 knock-in mouse model of ALS. Acta Neuropathol Commun. BioMed Central Ltd. 2020;8:3.
Ebstein SY, Yagudayeva I, Shneider NA. Mutant TDP-43 causes early-stage dose-dependent motor neuron degeneration in a TARDBP Knockin mouse model of ALS. Cell Rep. Elsevier. 2019;26:364–373.e4.
Mitchell JC, Constable R, So E, Vance C, Scotter E, Glover L, et al. Wild type human TDP-43 potentiates ALS-linked mutant TDP-43 driven progressive motor and cortical neuron degeneration with pathological features of ALS. Acta Neuropathol Commun. BioMed Central. 2015;3:36.
Hawrot J, Imhof S, Wainger BJ. Modeling cell-autonomous motor neuron phenotypes in ALS using iPSCs. Neurobiol Dis. Academic Press Inc. 2020;134:104680.
Bilican B, Serio A, Barmada SJ, Nishimura AL, Sullivan GJ, Carrasco M, et al. Mutant induced pluripotent stem cell lines recapitulate aspects of TDP-43 proteinopathies and reveal cell-specific vulnerability. Proc Natl Acad Sci U S A. National Academy of Sciences. 2012;109:5803–8.
Serio A, Bilican B, Barmada SJ, Ando DM, Zhao C, Siller R, et al. Astrocyte pathology and the absence of non-cell autonomy in an induced pluripotent stem cell model of TDP-43 proteinopathy. Proc Natl Acad Sci U S A. National Academy of Sciences. 2013;110:4697–702.
Zhang Z, Almeida S, Lu Y, Nishimura AL, Peng L, Sun D, et al. Downregulation of MicroRNA-9 in iPSC-derived neurons of FTD/ALS patients with TDP-43 mutations. Pandey U, editor. PLoS One. Public Library of Science. 2013;8:e76055.
Frank SA. Somatic evolutionary genomics: mutations during development cause highly variable genetic mosaicism with risk of cancer and neurodegeneration. Proc Natl Acad Sci U S A. National Academy of Sciences. 2010;107:1725–30.
Benatar M, Wuu J, Andersen PM, Lombardi V, Malaspina A. Neurofilament light: a candidate biomarker of presymptomatic amyotrophic lateral sclerosis and phenoconversion. Ann Neurol. John Wiley and Sons Inc. 2018;84:130–9.
Gisslén M, Price RW, Andreasson U, Norgren N, Nilsson S, Hagberg L, et al. Plasma concentration of the Neurofilament light protein (NFL) is a biomarker of CNS injury in HIV infection: a cross-sectional study. EBioMedicine. Elsevier B.V. 2016;3:135–40.
Li QF, Dong Y, Yang L, Xie JJ, Ma Y, Du YC, et al. Neurofilament light chain is a promising serum biomarker in spinocerebellar ataxia type 3. Mol Neurodegener. BioMed Central Ltd. 2019;14:39.
Preische O, Schultz SA, Apel A, Kuhle J, Kaeser SA, Barro C, et al. Serum neurofilament dynamics predicts neurodegeneration and clinical progression in presymptomatic Alzheimer’s disease. Nat Med. Nature Publishing Group. 2019;25:277–83.
Cai L, Huang J. Neurofilament light chain as a biological marker for multiple sclerosis: a meta-analysis study. Neuropsychiatr Dis Treat. Dove Medical Press Ltd. 2018;14:2241–54.
Gregory JM, McDade K, Livesey MR, Croy I, Marion de Proce S, Aitman T, et al. Spatial transcriptomics identifies spatially dysregulated expression of GRM3 and USP47 in amyotrophic lateral sclerosis. Neuropathol Appl Neurobiol. Blackwell Publishing Ltd. 2020. https://doi.org/10.1111/nan.12597.
Maniatis S, Äijö T, Vickovic S, Braine C, Kang K, Mollbrink A, et al. Spatiotemporal dynamics of molecular pathology in amyotrophic lateral sclerosis. Science. American Association for the Advancement of Science. 2019;364:89–93.
Otake K, Kamiguchi H, Hirozane Y. Identification of biomarkers for amyotrophic lateral sclerosis by comprehensive analysis of exosomal mRNAs in human cerebrospinal fluid. BMC Med Genomics. BioMed Central Ltd. 2019;12:7.
Zucca S, Gagliardi S, Pandini C, Diamanti L, Bordoni M, Sproviero D, et al. RNA-seq profiling in peripheral blood mononuclear cells of amyotrophic lateral sclerosis patients and controls. Sci Data. Nature Publishing Groups. 2019;6:1–8.
Tyzack GE, Luisier R, Taha DM, Neeves J, Modic M, Mitchell JS, et al. Widespread FUS mislocalization is a molecular hallmark of amyotrophic lateral sclerosis. Brain. 2019;142(9):2572–80.
Lobingier BT, Hüttenhain R, Eichel K, Miller KB, Ting AY, von Zastrow M, et al. An approach to spatiotemporally resolve protein interaction networks in living cells. Cell. 2017;169:350–360.e12.
Roux KJ, Kim DI, Burke B. BioID: A screen for protein-protein interactions. Curr Protoc Protein Sci. Blackwell Publishing Inc. 2013;2013:19.23.1–19.23.14.
Fazal FM, Han S, Parker KR, Kaewsapsak P, Xu J, Boettiger AN, et al. Atlas of subcellular RNA localization revealed by APEX-Seq. Cell. Elsevier. 2019;178:1–18.
Padrón A, Iwasaki S, Ingolia NT. Proximity RNA Labeling by APEX-Seq Reveals the Organization of Translation Initiation Complexes and Repressive RNA Granules. Mol Cell. Cell Press. 2019;75:875–887.e5.
Markmiller S, Soltanieh S, Server KL, Bennett EJ, Lé E, Yeo Correspondence GW. Context-Dependent and Disease-Specific Diversity in Protein Interactions within Stress Granules. Cell. 2018;172:590–598.e13.
Ule J, Jensen KB, Ruggiu M, Mele A, Ule A, Darnell RB. CLIP Identifies Nova-Regulated RNA Networks in the brain. Science. American Association for the Advancement of Science. 2003;302:1212–5.
Schwab C, Arai T, Hasegawa M, Yu S, McGeer PL. Colocalization of transactivation-responsive DNA-binding protein 43 and huntingtin in inclusions of Huntington disease. J Neuropathol Exp Neurol. Oxford Academic. 2008;67:1159–65.
Nakashima-Yasuda H, Uryu K, Robinson J, Xie SX, Hurtig H, Duda JE, et al. Co-morbidity of TDP-43 proteinopathy in Lewy body related diseases. Acta Neuropathol. 2007;114:221–9.
Chornenkyy Y, Fardo DW, Nelson PT. Tau and TDP-43 proteinopathies: kindred pathologic cascades and genetic pleiotropy. Lab Investig. Nature Publishing Group. 2019;99:993–1007.
Nonaka T, Masuda-Suzukake M, Hosokawa M, Shimozawa A, Hirai S, Okado H, et al. C9ORF72 dipeptide repeat poly-GA inclusions promote intracellular aggregation of phosphorylated TDP-43. Hum Mol Genet. 2018;27:2658–70.
Acknowledgments
Figures were created with Biorender.com
Funding
T.R.S. acknowledges the ALS Society of Canada in partnership with the Brain Canada Foundation through the Brain Canada Research Fund, with the financial support of Health Canada, in addition to the Éric Poulin Center for Neuromuscular Disease for financial support through the ALS Trainee Award Program 2019 and Scholarship in Translational Research awards, respectively. M.W.C.R. acknowledges the Parkinson’s Foundation (PF-JFA-1762), Parkinson’s Canada (New Investigator Award), the New Frontiers in Research Fund (NFRFE-2018-00264), Brain Canada-Azrieli Foundation (Early-Career Capacity Building Grant) and Partners Invested in Parkinson’s Research (PIPR). None of these funding sources played a role in writing or editing of this manuscript and the comments in this manuscript solely represent those of the authors. The views expressed herein do not necessarily represent the views of the Minister of Health or the Government of Canada.
Author information
Authors and Affiliations
Contributions
T.R.S. and M.W.C.R. wrote and edited the manuscript. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Suk, T.R., Rousseaux, M.W.C. The role of TDP-43 mislocalization in amyotrophic lateral sclerosis. Mol Neurodegeneration 15, 45 (2020). https://doi.org/10.1186/s13024-020-00397-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13024-020-00397-1